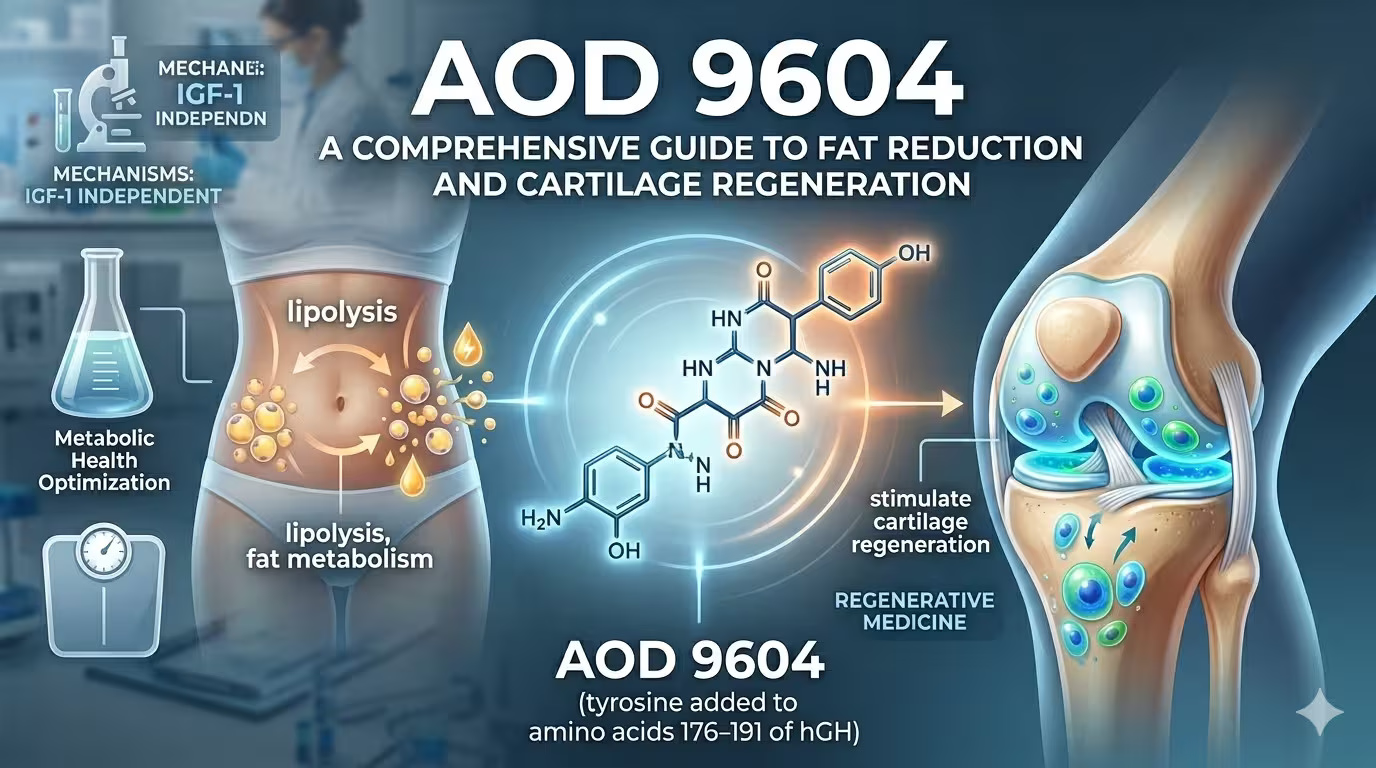
Among the growing landscape of bioactive peptides used in metabolic and regenerative medicine, AOD 9604 occupies a unique position. Derived from the C-terminal fragment of human growth hormone (hGH), this 16-amino-acid synthetic peptide was engineered to isolate the fat-metabolizing properties of growth hormone — without triggering its systemic hormonal side effects. What began as an anti-obesity drug candidate in the 1990s has evolved into a versatile therapeutic tool with applications spanning targeted fat reduction, cartilage regeneration, post-surgical recovery, and metabolic health optimization.
This guide synthesizes over two decades of peer-reviewed research, clinical trial data from six randomized controlled studies involving 925 patients, and real-world clinical observations from Dr. Adam Sewell, M.D. — a medical entrepreneur and peptide therapy specialist (@medicalentrepreneur) — to provide the most comprehensive overview of AOD 9604 available for researchers and clinicians.
What Is AOD 9604? Molecular Origins and Design
AOD 9604 (Anti-Obesity Drug 9604) is a synthetic peptide corresponding to amino acids 176–191 of the human growth hormone molecule, with an additional tyrosine residue at the N-terminus for structural stability. It was originally developed in the 1990s by Dr. Frank Ng and colleagues at Monash University in Melbourne, Australia, through Metabolic Pharmaceuticals Ltd.
The foundational premise was elegant: human growth hormone is a 191-amino-acid polypeptide with diverse biological functions — growth promotion, glucose regulation, and lipid metabolism among them. Researchers identified that the lipolytic activity of hGH was concentrated in the C-terminal region (residues 177–191), while the growth-promoting and diabetogenic effects were mediated through different structural domains and the IGF-1 signaling pathway [1]. By isolating this specific fragment and modifying it into AOD 9604, scientists created a peptide that could stimulate fat breakdown without activating hGH receptors or elevating IGF-1 levels [2].
This distinction is critical. Full-length growth hormone therapy, while effective for body recomposition, carries well-documented risks including insulin resistance, glucose intolerance, fluid retention, and potential tumor growth stimulation. AOD 9604 was specifically designed to bypass these pathways, offering a targeted metabolic intervention with a substantially improved safety profile.
Mechanism of Action: How AOD 9604 Works
AOD 9604 exerts its biological effects through two primary and distinct mechanisms — lipid metabolism modulation and tissue repair signaling — neither of which involves direct interaction with growth hormone receptors.
1. Fat Metabolism: Lipolysis Activation and Lipogenesis Inhibition
The peptide's primary mechanism centers on adipose tissue, where it produces a dual action:
Stimulation of lipolysis — AOD 9604 activates hormone-sensitive lipase (HSL) in adipocytes, the enzyme responsible for breaking down stored triglycerides into free fatty acids and glycerol for energy utilization. This process is particularly effective in visceral and abdominal fat depots, the most metabolically dangerous fat stores associated with cardiovascular disease and insulin resistance.
Inhibition of lipogenesis — Simultaneously, the peptide suppresses acetyl-CoA carboxylase, a key enzyme in the de novo fatty acid synthesis pathway. This prevents the formation of new fat from circulating substrates, creating a net-negative fat balance when combined with appropriate caloric management.
Research by Heffernan et al. (2001) demonstrated that this dual mechanism is partially mediated through upregulation of β3-adrenergic receptor (β3-AR) expression in adipose tissue. In obese mice, both hGH and AOD 9604 restored suppressed β3-AR RNA levels to those comparable with lean mice, correlating with significant reductions in body weight and white adipose tissue mass after 14 days of treatment [2]. Importantly, while long-term lipolytic effects required β3-AR (as confirmed in knockout mice), acute administration of AOD 9604 still increased energy expenditure and fat oxidation even in β3-AR knockout models — suggesting the existence of additional, β3-AR-independent pathways [2].
2. Cartilage Repair and Tissue Regeneration
A second, more recently explored mechanism positions AOD 9604 as a regenerative peptide. Research by Kwon and Park (2015) demonstrated that intra-articular injection of AOD 9604 in a collagenase-induced osteoarthritis rabbit model enhanced cartilage regeneration, with histopathological analysis showing superior outcomes in cartilage surface integrity and reduced chondrocyte disorganization [4]. When combined with hyaluronic acid (HA), the effects were synergistic — the combined injection group showed significantly lower cartilage degeneration scores and shorter lameness periods compared to either treatment alone [4].
The proposed mechanism involves AOD 9604's ability to recapitulate developmental growth cascades in articular cartilage, promoting chondrocyte proliferation and extracellular matrix production without the systemic effects associated with full-length growth hormone therapy. This has positioned AOD 9604 among a class of peptides — including BPC-157 and Thymosin β4 (TB-500) — recognized for their potential in musculoskeletal regeneration [7].
3. Metabolic Safety: The IGF-1 Independence
Perhaps the most clinically significant aspect of AOD 9604's mechanism is what it does not do. Across six randomized, double-blind, placebo-controlled clinical trials, AOD 9604 demonstrated no effect on serum IGF-1 levels, no impact on insulin sensitivity or glucose tolerance (as assessed by oral glucose tolerance testing), and no stimulation of anti-AOD 9604 antibody formation [3]. This confirms that the peptide operates entirely outside the hGH/IGF-1 axis, offering fat-metabolizing benefits without the metabolic risks of growth hormone therapy.
Clinical Evidence: From Preclinical Studies to Human Trials
Preclinical Research
The foundational preclinical work was conducted at Monash University. In 2000, Ng et al. published seminal findings showing that oral administration of AOD 9604 (500 µg/kg/day for 19 days) in obese Zucker rats reduced body weight gain by over 50% (15.8 ± 0.6 g vs. 35.6 ± 0.8 g in controls), increased lipolytic activity in adipose tissue, and — critically — produced no adverse effects on insulin sensitivity as measured by euglycemic clamp techniques [1].
Heffernan et al. (2000) further demonstrated that oral AOD 9604 administration increased fat oxidation in both lean and obese mice, with chronic treatment reducing white adipose tissue mass and epididymal fat pad weight without altering caloric intake — indicating a metabolic rather than anorectic mechanism [5].
Human Clinical Trials
Over $50 million was invested in the clinical development of AOD 9604, resulting in six randomized, double-blind, placebo-controlled clinical trials conducted in the United Kingdom and Australia between 2001 and 2007, involving a total of 925 patients [3].
The key findings from the human trial program include:
12-week efficacy trial (METAOD005) — Subjects receiving AOD 9604 at 1 mg/day orally demonstrated an average weight reduction of 2.6 kg compared to 0.8 kg in the placebo group, representing a statistically significant difference in a relatively short treatment window [6].
24-week pivotal trial (METAOD006) — This larger, multi-center study enrolled 534 clinically obese subjects (BMI 30–45 kg/m²) across 16 Australian hospitals. Participants were randomized to receive 0.25, 0.5, or 1 mg AOD 9604 or placebo daily for 24 weeks. While the primary endpoint of significant weight loss was not met — leading to discontinuation of the oral drug development program in 2007 — the study provided extensive safety data demonstrating an excellent tolerability profile [3][6].
Safety across all trials — Analysis of safety data from all six trials confirmed that AOD 9604 had no clinically relevant effects on IGF-1 levels, no negative impact on glucose metabolism, no immunogenic response (anti-AOD 9604 antibodies were not detected in any subject at any timepoint), and no symptoms associated with IGF-1-mediated effects such as sodium retention, tissue edema, or hypertension [3].
It is important to note that the failure of the 24-week oral trial to meet its primary weight-loss endpoint does not invalidate the peptide's biological activity. The oral bioavailability of peptides is inherently limited, and current clinical protocols predominantly use subcutaneous injection, which provides substantially higher bioavailability and more consistent tissue-level concentrations than oral formulations.
Clinical Applications in Modern Practice
Based on the available evidence and clinical experience reported by practitioners including Dr. Adam Sewell, M.D., AOD 9604 is currently utilized across several therapeutic domains:
Targeted Fat Reduction
The primary application remains body fat reduction, particularly for patients with stubborn visceral and abdominal adiposity that has proven resistant to diet and exercise interventions. AOD 9604 is particularly valuable for patients with metabolic syndrome, where the selective fat-mobilizing action without insulin resistance is a critical advantage over alternative therapies.
Metabolic Syndrome Support
For patients with insulin resistance, elevated triglycerides, and central obesity — the hallmarks of metabolic syndrome — AOD 9604 offers a therapeutic pathway that addresses fat accumulation without exacerbating glucose dysregulation. This metabolic neutrality distinguishes it from growth hormone therapy and makes it suitable for a population that might otherwise be contraindicated for hGH-based interventions.
Osteoarthritis and Cartilage Regeneration
Emerging evidence supports AOD 9604 for non-surgical management of osteoarthritis, particularly when combined with hyaluronic acid. The rabbit model data from Kwon and Park showing enhanced cartilage regeneration [4], combined with clinical observations of improved joint function and reduced pain scores, has established a growing role for AOD 9604 in regenerative orthopedics.
Post-Surgical Recovery
The tissue repair properties of AOD 9604 extend to post-surgical rehabilitation, where the peptide may accelerate healing timelines in orthopedic and musculoskeletal procedures. The enhancement of cellular repair processes, combined with anti-inflammatory signaling, supports faster recovery of range of motion and functional strength.
Body Recomposition and Anti-Aging
When integrated into comprehensive wellness protocols that include structured exercise and nutritional optimization, AOD 9604 supports improvements in the lean-mass-to-fat ratio. For aging populations experiencing age-related metabolic decline and increased adiposity, the peptide offers metabolic support without the risks associated with hormone replacement.
Dosage Protocols and Administration
AOD 9604 is administered via subcutaneous injection, typically into abdominal adipose tissue for optimal absorption. Based on clinical trial data and practitioner experience, the following protocols are established:
Fat Reduction Protocol: 200–300 mcg administered subcutaneously once daily, preferably in the morning on an empty stomach. This aligns with circadian fat metabolism patterns and clinical trial dosing that demonstrated optimal lipolytic activity without diminishing returns at higher doses.
Cartilage and Tissue Repair Protocol: 250–500 mcg administered subcutaneously daily or every other day, depending on the severity of the condition and clinical response. For osteoarthritis applications, some practitioners use this in conjunction with intra-articular hyaluronic acid injections based on the synergistic data from preclinical models.
Post-Surgical Recovery Protocol: 300–500 mcg administered subcutaneously daily for 4–6 weeks post-procedure, with the goal of accelerating tissue repair and reducing recovery timelines.
Treatment duration typically ranges from 4 to 12 weeks depending on the clinical objective and patient response. Cycling protocols (8 weeks on, 4 weeks off) are commonly employed to maintain peptide sensitivity, with periodic reassessment of outcomes and biomarkers.
Laboratory Monitoring and Patient Assessment
While AOD 9604 does not impact systemic hormone levels, comprehensive laboratory monitoring is recommended to ensure patient safety and track therapeutic progress:
Baseline Assessment: Comprehensive metabolic panel (CMP) to evaluate kidney and liver function, fasting glucose and insulin levels; complete lipid panel including triglycerides, LDL, HDL, and VLDL; body composition measurements (DEXA scan or bioimpedance analysis); and inflammatory markers (C-reactive protein, ESR) for patients undergoing tissue repair protocols.
During Treatment: Periodic body weight and body fat percentage measurements at 4-week intervals; patient-reported outcomes regarding joint pain, mobility, and tissue healing for regenerative protocols; metabolic markers reassessment at the midpoint of treatment.
Post-Treatment Evaluation: Complete metabolic and body composition reassessment to evaluate treatment efficacy; advanced imaging (MRI or ultrasound) for patients undergoing cartilage or tissue repair to assess structural improvements objectively.
Real-World Clinical Case Studies
The following case studies, presented by Dr. Adam Sewell, M.D. (@medicalentrepreneur), illustrate the practical application and outcomes of AOD 9604 across different clinical scenarios:
Case Study 1: Abdominal Fat Reduction in Metabolic Syndrome
Patient Profile: A 45-year-old male presenting with metabolic syndrome, characterized by central obesity, elevated triglycerides, and borderline fasting glucose. Despite consistent adherence to caloric restriction and regular exercise over 6 months, stubborn abdominal fat deposits remained resistant to lifestyle intervention alone.
Treatment Protocol: AOD 9604 at 300 mcg administered subcutaneously once daily for 12 weeks, in conjunction with continued dietary management and structured physical activity.
Outcome: The patient achieved a 15% reduction in abdominal fat as measured by serial body composition analysis. Concurrent laboratory improvements included meaningful reductions in fasting triglyceride levels and fasting glucose concentrations, bringing both markers closer to optimal reference ranges. No adverse effects were reported throughout the 12-week treatment period, and the improvements in metabolic markers were sustained at 8-week follow-up.
Case Study 2: Cartilage Repair in Knee Osteoarthritis
Patient Profile: A 55-year-old male with moderate knee osteoarthritis confirmed by imaging, experiencing chronic pain and progressive limitation of mobility. The patient sought non-surgical alternatives to delay or avoid total knee replacement.
Treatment Protocol: AOD 9604 at 400 mcg administered subcutaneously once daily for 8 weeks, combined with physical therapy and joint-protective supplementation.
Outcome: The patient reported significant reduction in knee pain scores and measurable improvement in joint mobility by week 8. Follow-up imaging demonstrated evidence of cartilage improvement consistent with regenerative processes. These findings align with the preclinical data from Kwon and Park (2015) showing enhanced cartilage regeneration with AOD 9604 in osteoarthritis models [4]. The patient was able to resume moderate physical activities that had previously been limited by joint pain.
Case Study 3: Post-Surgical Recovery After ACL Reconstruction
Patient Profile: A 30-year-old competitive athlete recovering from anterior cruciate ligament (ACL) reconstruction surgery, motivated to optimize recovery timelines and return to training as quickly and safely as possible.
Treatment Protocol: AOD 9604 at 500 mcg administered subcutaneously once daily for 6 weeks, initiated during the early rehabilitation phase alongside a structured physical therapy program.
Outcome: The patient demonstrated accelerated recovery of range of motion and functional strength compared to typical rehabilitation timelines for ACL reconstruction. Physical therapy milestones were achieved ahead of schedule, and the patient reported fewer adverse symptoms (swelling, stiffness) during the recovery process. No treatment-related side effects were observed, and the athlete was cleared for progressive return to sport-specific training earlier than the standard rehabilitation timeline would predict.
Safety Profile and Risk Considerations
Based on the totality of evidence from six clinical trials involving 925 patients, extensive preclinical safety testing, and clinical practice observations, AOD 9604 demonstrates what peer-reviewed literature describes as a very good safety and tolerability profile [3].
Confirmed safety findings include: no effect on serum IGF-1 levels (confirming independence from the hGH/IGF-1 axis); no negative impact on glucose metabolism or insulin sensitivity; no immunogenic response (no anti-AOD 9604 antibodies detected); no clinically significant changes in blood pressure, cholesterol, or hematological parameters; and no evidence of genotoxicological or toxicological concerns in non-clinical safety studies [3].
Potential minor side effects reported in clinical settings include mild injection-site reactions (redness, transient swelling), occasional mild headache, and rare instances of transient nausea. These effects are generally self-limiting and resolve without intervention.
Important considerations: AOD 9604 is classified as a prohibited substance by the World Anti-Doping Agency (WADA) and should not be used by competitive athletes subject to anti-doping regulations. Additionally, while short-to-medium-term safety data is reassuring, long-term safety studies beyond 24 weeks in humans remain limited, and clinicians should exercise appropriate clinical judgment regarding extended treatment protocols.
Therapeutic Alternatives and Comparisons
AOD 9604 exists within a broader therapeutic landscape, and understanding its relative positioning helps clinicians make informed decisions:
For fat reduction: GLP-1 receptor agonists (GLP-1 agonist peptide, liraglutide) offer stronger weight-loss outcomes through appetite suppression but carry gastrointestinal side effects and operate through fundamentally different pathways. L-carnitine and lipotropic injections provide milder metabolic support. AOD 9604's niche is targeted lipolysis without appetite alteration or hormonal disruption.
For cartilage repair: Platelet-rich plasma (PRP) and hyaluronic acid injections are established therapies, and the preclinical evidence suggests AOD 9604 combined with HA may offer superior outcomes to either alone [4]. BPC-157 and TB-500 are complementary peptides with overlapping regenerative mechanisms that are sometimes used in combination protocols.
For tissue healing: BPC-157 (body protection compound) targets gastrointestinal and tendon healing, while TB-500 (Thymosin Beta-4) promotes systemic tissue repair through different molecular pathways. AOD 9604 complements these through its growth-factor-like regenerative signaling in cartilage and musculoskeletal tissue.
Conclusion
AOD 9604 represents a thoughtfully engineered therapeutic peptide that successfully isolates the metabolic benefits of human growth hormone from its systemic risks. With over $50 million in clinical development, six controlled human trials, and a growing body of regenerative medicine research, it has established itself as a versatile tool in modern peptide therapy.
Its dual mechanism — activating lipolysis while inhibiting lipogenesis in adipose tissue, and promoting cartilage regeneration in articular joints — combined with its confirmed independence from the IGF-1 pathway and excellent safety profile, makes AOD 9604 a valuable option for clinicians managing stubborn adiposity, metabolic syndrome, osteoarthritis, and post-surgical rehabilitation.
As with all peptide therapies, optimal outcomes depend on proper patient selection, individualized dosing, comprehensive laboratory monitoring, and integration within a broader clinical strategy that includes nutrition, physical activity, and ongoing medical oversight. The clinical case studies presented by Dr. Adam Sewell, M.D. demonstrate the real-world applicability of these protocols across diverse patient populations and therapeutic goals.
This article is intended for educational and research purposes. AOD 9604 is not FDA-approved for therapeutic use. Consult with a qualified healthcare provider before initiating any peptide therapy protocol.