BPC-157 Peptide
Pentadecapeptide (15 amino acids) derived from human gastric juice protein. Extensively researched for tissue repair, angiogenesis, wound healing, tendon recovery, gastrointestinal protection, and neuroprotective mechanisms. One of the most studied regenerative peptides in preclinical literature.
| Simple Peptide | GoAlpha Labs | AminoCore ✓ all‑in | |
|---|---|---|---|
| 10mg price | $55.00 | $35.99 | $55.00 |
| Shipping | +$13.00 | +$13.00 | FREE |
| Bac Water | +$30.00 | +$15.00 | FREE |
| Total paid | $98.00 | $63.99 | $55.00 save up to $43.00 |
Quick Facts
| SKU | ACR-BPC157 |
|---|---|
| CAS Number | 137525-51-0 |
| Molecular Formula | C62H98N16O22 |
| Molecular Weight | 1419.53 g/mol |
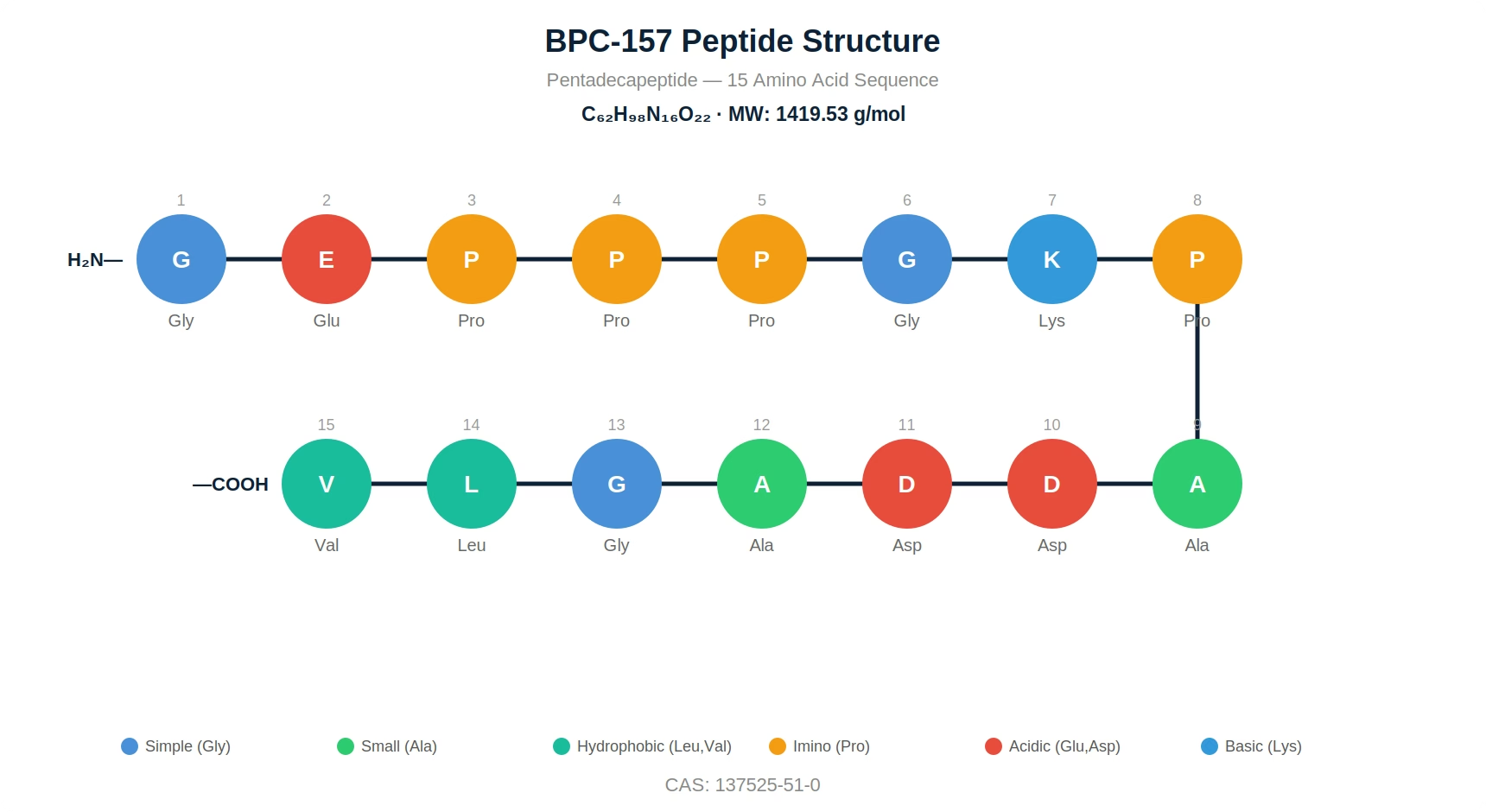
| Sequence | Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val |
| Purity | ≥99% |
| Physical Form | Lyophilized Powder |
| Storage | Store at -20°C |
What is BPC-157?
BPC-157 (Body Protection Compound-157) is a synthetic 15-amino-acid pentadecapeptide, sequence Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val, molecular weight 1,419.53 g/mol, CAS 137525-51-0, derived from a partial sequence of a cytoprotective protein isolated from human gastric juice. Published preclinical research describes activity in wound healing, tendon and ligament repair, gastrointestinal mucosal protection, and angiogenesis — primarily via FAK-paxillin pathway activation, nitric oxide homeostasis, and VEGF upregulation. AminoCore Research supplies BPC-157 at ≥98% HPLC purity, lyophilized, with per-lot Certificate of Analysis, for laboratory research use only.
BPC-157 (Body Protection Compound-157) is a synthetic pentadecapeptide consisting of 15 amino acids (Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val) with a molecular weight of 1,419.53 g/mol and CAS number 137525-51-0. It is derived from a partial sequence of the human gastric juice protein known as Body Protection Compound (BPC) (see What is BPC-157?), originally isolated and characterized by Professor Predrag Sikiric at the University of Zagreb.
BPC-157 has been the subject of over 100 preclinical research publications (BPC-157 research guide), making it one of the most extensively studied regenerative peptides in the scientific literature. Key research areas include wound healing, tendon and ligament repair, gastrointestinal mucosal protection, angiogenesis, and neuroprotection. Notably, BPC-157 demonstrates stability in human gastric juice — a unique property among peptides that typically undergo rapid degradation in acidic environments (stable at pH 1.0 for >24 hours).
Unlike most bioactive peptides, BPC-157 does not interact with growth hormone, somatostatin, or other classical endocrine axes. Its mechanisms appear to be primarily mediated through nitric oxide (NO) signaling, FAK-paxillin pathway activation, and growth factor modulation (VEGF, FGF-2, EGF), positioning it as a fundamentally different class of research compound from growth hormone secretagogues or receptor agonists. Detailed pathway diagrams are available in the companion article on BPC-157 mechanism of action.
Related AminoCore blends featuring BPC-157: WOLVERINE (with TB-500), GLOW (with TB-500 and GHK-Cu), KLOW (with TB-500, GHK-Cu, and KPV), and GLOW+ (with thymosin α-1). See the KLOW blend article for combinatorial rationale.
Mechanism of Action
Research & Clinical Studies
BPC-157 and Wound Healing Research
BPC-157 and Tendon Healing Research
[3] Chang CH, et al. The promoting effect of pentadecapeptide BPC 157 on tendon healing. J Appl Physiol. 2011;110(3):774-80. PubMed ↗
BPC-157 and Gastrointestinal Research
BPC-157 and Muscle Healing Research
[6] Pevec D, et al. Impact of pentadecapeptide BPC 157 on muscle healing impaired by systemic corticosteroid application. Med Sci Monit. 2010;16(3):BR81-88. PubMed ↗
BPC-157 and Central Nervous System Research
Chemical & Physical Properties
BPC-157 is a pentadecapeptide with the following verified specifications:
| Full Name | Body Protection Compound-157 |
|---|---|
| Molecular Formula | C₆₂H₉₈N₁₆O₂₂ |
| Molecular Weight | 1,419.53 g/mol (free acid form) |
| CAS Number | 137525-51-0 |
| Sequence | Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val |
| Amino Acids | 15 (pentadecapeptide) |
| Origin | Partial sequence of human gastric juice protein BPC |
| Physical Form | White to off-white lyophilized powder |
| Solubility | Freely soluble in water and bacteriostatic water |
| Gastric Stability | Stable at pH 1.0 for >24 hours (unique for peptides) |
| Purity | ≥98% by HPLC |
| Isoelectric Point | ~4.2 (acidic peptide) |
BPC-157 is available in both free acid and acetate salt forms. The free acid form (CAS 137525-51-0) is the more commonly used form in research. The peptide contains three proline residues (positions 3-5) that contribute to its unusual stability and resistance to proteolytic degradation.
Handling & Reconstitution Guidelines
Reconstitution Protocol:
- Allow the sealed BPC-157 vial and bacteriostatic water to reach room temperature (15-25°C).
- Swab both vial stoppers with sterile alcohol prep pads. Allow to air dry.
- Using a sterile insulin syringe, withdraw the desired volume of bacteriostatic water.
- Insert the needle into the BPC-157 vial at an angle, directing the water stream gently down the inside wall — never directly onto the lyophilized powder.
- Allow the vial to sit for 2-5 minutes. BPC-157 dissolves readily due to its hydrophilic nature.
- If needed, gently roll the vial between palms. Do not shake or vortex.
- The solution should be clear and colorless. Discard if cloudy, discolored, or containing particles.
Recommended Reconstitution: For a 5 mg vial, adding 1 mL of BAC water yields a 5 mg/mL concentration (250 mcg per 0.05 mL). For 10 mg vials, 2 mL yields the same concentration.
Note: BPC-157 is unusually stable for a peptide — it withstands acidic conditions (pH 1.0) and is resistant to proteolytic enzymes. This makes it one of the easier research peptides to handle. However, standard cold-chain practices still maximize shelf life.
Storage & Stability Information
Lyophilized (unreconstituted):
- Long-term: -20°C for up to 24 months
- Short-term: 2-8°C for up to 6 months
- Room temperature: Stable for up to 30 days (transit)
- Protect from light and moisture
Reconstituted:
- Refrigerate at 2-8°C immediately after reconstitution
- Use within 21 days when stored at 2-8°C
- Do not freeze reconstituted solution
- Avoid repeated freeze-thaw cycles
Stability Advantage: BPC-157 is significantly more stable than most research peptides due to its proline-rich sequence and acidic isoelectric point. In gastric juice conditions (pH 1.0-2.0), BPC-157 remains active for >24 hours — a stability profile unmatched by other peptides of similar size. This stability extends to reconstituted solutions, where BPC-157 maintains potency longer than GH secretagogues or GLP-1 analogs.
Frequently Asked Questions
What is BPC-157?
BPC-157 (Body Protection Compound-157) is a synthetic pentadecapeptide consisting of 15 amino acids derived from a partial sequence of a protein found in human gastric juice. It has been extensively studied in preclinical research for its interactions with various biological pathways including tissue repair, angiogenesis, and neuroprotection. BPC-157 is available exclusively for laboratory and research purposes.
What is the molecular weight and formula of BPC-157?
BPC-157 has a molecular formula of C62H98N16O22 and a molecular weight of 1419.53 g/mol. Its amino acid sequence is Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val. The CAS registry number is 137525-51-0.
How should BPC-157 be stored?
Lyophilized BPC-157 should be stored at -20C in a sealed container protected from light and moisture, where it maintains stability for up to 24 months. After reconstitution, store at 2-8C and use within 30 days.
What purity level is available for BPC-157?
AminoCore Research provides BPC-157 at 98% purity or higher as verified by High-Performance Liquid Chromatography (HPLC). Each batch is accompanied by a Certificate of Analysis (COA) documenting the purity testing results.
How is BPC-157 reconstituted for laboratory use?
BPC-157 lyophilized powder should be reconstituted using sterile bacteriostatic water or saline solution. Allow the vial to reach room temperature, inject the diluent slowly along the vial wall, and gently swirl until dissolved. A typical reconstitution volume is 1-2 mL per 5mg vial.
What areas of research has BPC-157 been studied in?
Published preclinical research on BPC-157 spans multiple areas including wound healing and tissue repair, tendon and ligament recovery, gastrointestinal function, muscle healing under corticosteroid conditions, neuroprotection and traumatic brain injury, and interactions with the dopaminergic system.
Is BPC-157 stable in acidic conditions?
Yes, BPC-157 is noted in published literature for its remarkable stability in gastric juice conditions, which distinguishes it from many other peptides that undergo rapid degradation in acidic environments.
What is the difference between BPC-157 and TB-500?
BPC-157 and TB-500 (Thymosin Beta-4) are both research peptides studied for their roles in tissue biology, but they operate through different mechanisms. BPC-157 is a 15-amino acid peptide that interacts with the NO system and FAK-paxillin pathway. TB-500 is a 43-amino acid peptide that primarily acts as an actin-sequestering molecule involved in cell motility.
Can BPC-157 be combined with TB-500 in research studies?
BPC-157 and TB-500 have distinct but convergent mechanisms — BPC-157 is associated with FAK-paxillin and nitric oxide signaling, while TB-500 is associated with G-actin sequestration and cytoskeletal remodeling. AminoCore supplies pre-combined research blends for investigators studying these pathways in parallel: WOLVERINE (two peptides), GLOW (three peptides), and KLOW (four peptides). The direct comparison is summarized in BPC-157 vs TB-500.
For laboratory and research use only. Not intended for human or animal consumption. All product information is derived from published preclinical research and does not constitute medical advice or claims.