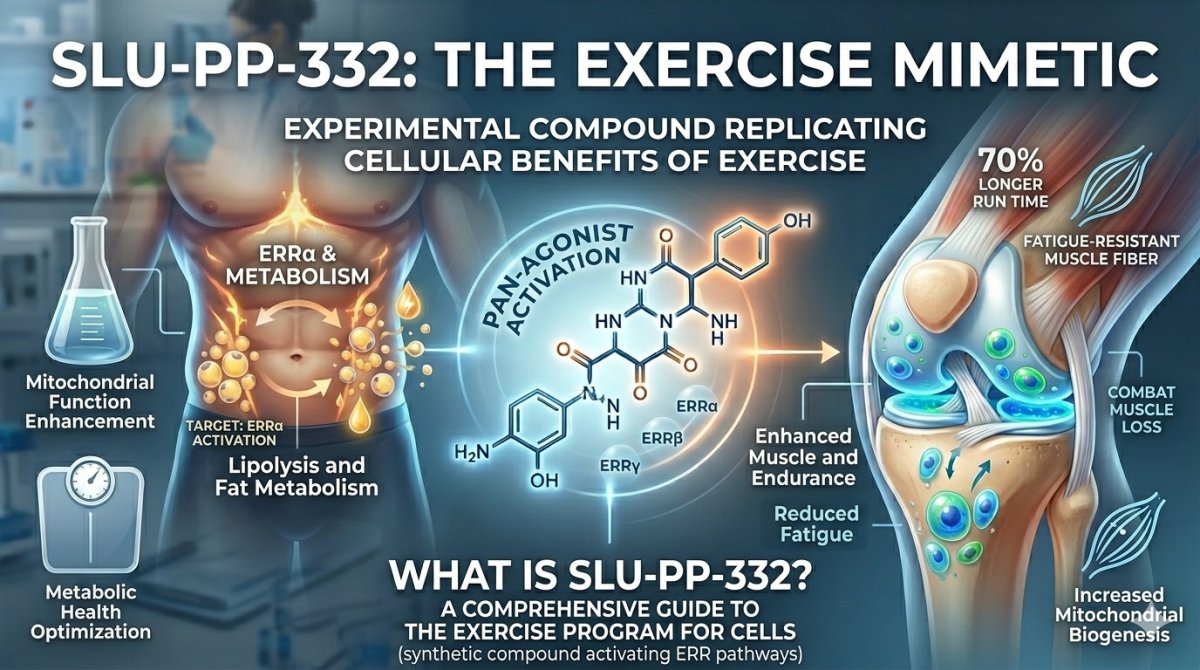
Among the emerging class of compounds known as exercise mimetics, SLU-PP-332 has attracted significant attention from researchers in metabolic medicine, cardiovascular science, and aging biology. Developed at Saint Louis University by the research group led by Thomas P. Burris, Ph.D. and Cyrielle Billon, Ph.D., this synthetic small molecule activates the estrogen-related receptor (ERR) family of nuclear receptors — triggering transcriptional programs that govern mitochondrial biogenesis, fatty acid oxidation, and cellular respiration. The result is a molecular signature that closely resembles the cellular adaptations produced by sustained aerobic exercise, without physical exertion.
This guide synthesizes the available peer-reviewed literature — spanning publications in ACS Chemical Biology, Circulation, Journal of Pharmacology and Experimental Therapeutics, American Journal of Pathology, and Frontiers in Physiology — to provide a rigorous overview of SLU-PP-332's pharmacology, preclinical efficacy, therapeutic potential, and current limitations.
Clarifying a Widespread Misidentification
Before examining the science, it is important to correct a factual error that has propagated through peptide therapy communities and supplement-oriented websites. SLU-PP-332 is not a peptide. It is a small molecule — specifically, a hydrazide derivative with the chemical name (E)-4-Hydroxy-N'-(naphthalen-2-ylmethylene)benzohydrazide and CAS number 303760-60-3. The designation "SLU-PP" refers to the compound series originating from Saint Louis University, not "Selective Lipid Uptake Peptide" as some sources have incorrectly stated.
This distinction carries practical significance. The pharmacokinetic profile, oral bioavailability, receptor binding dynamics, and storage requirements of a small molecule differ fundamentally from those of peptide therapeutics. Misclassifying SLU-PP-332 as a peptide leads to incorrect assumptions about dosing, administration route, stability, and mechanism — all of which can compromise both research integrity and clinical reasoning.
Pharmacological Profile and Receptor Selectivity
SLU-PP-332 functions as a pan-agonist of the three estrogen-related receptors: ERRα, ERRβ, and ERRγ. Despite the name, these orphan nuclear receptors are not involved in estrogen signaling. They are transcription factors that regulate gene networks controlling energy metabolism, mitochondrial function, and cellular respiration. The compound demonstrates the following binding affinities:
ERRα — EC50 of 98 nM, with primary expression in skeletal muscle, heart, and brown adipose tissue. This is the principal mediator of the compound's exercise-mimetic effects.
ERRβ — EC50 of 230 nM, with primary expression in brain, kidney, and retinal tissue.
ERRγ — EC50 of 430 nM, with primary expression in heart, kidney, and skeletal muscle. This receptor plays a central role in the compound's cardioprotective effects.
When activated, ERRα drives the expression of hundreds of genes associated with aerobic fitness. Key downstream targets include PGC-1α (the master regulator of mitochondrial biogenesis), GLUT4 (a key insulin- and exercise-responsive glucose transporter), uncoupling proteins (UCPs) involved in thermogenesis and energy expenditure, and a suite of fatty acid oxidation enzymes including CPT1 and MCAD [1]. The net result is a cellular environment that preferentially oxidizes fatty acids for energy, increases mitochondrial density, and enhances oxidative phosphorylation capacity — changes that normally require sustained endurance training to achieve.
Mechanism of Action: How SLU-PP-332 Works
SLU-PP-332 exerts its biological effects through a single, well-characterized mechanism — activation of ERR nuclear receptors — that produces diverse downstream effects across multiple organ systems.
1. Exercise-Mimetic Transcriptional Reprogramming
The compound's primary mechanism centers on ERRα activation in energy-demanding tissues, particularly skeletal muscle. Upon binding, SLU-PP-332 induces an ERRα-dependent acute aerobic exercise genetic program — a coordinated activation of gene networks that normally require physical exercise to engage. This program shifts cellular fuel preference from glucose toward fatty acids, increases mitochondrial biogenesis and respiratory capacity, and promotes the formation of oxidative (Type IIa) muscle fibers associated with endurance performance [1].
Critically, researchers confirmed that this effect is specifically ERRα-dependent: when ERRα signaling was experimentally blocked, the exercise-enhancing effects were abolished. This established that ERRα activation alone is sufficient to induce the acute exercise transcriptional program, providing a clear mechanistic basis for the compound's classification as an exercise mimetic [1].
2. Metabolic Fuel Switching
The transcriptional changes induced by SLU-PP-332 produce a measurable shift in whole-body fuel metabolism. Treated animals demonstrated a reduced respiratory exchange ratio (RER) — a direct indicator that the body is oxidizing a higher proportion of fatty acids relative to carbohydrates. This metabolic reprogramming increases energy expenditure and fat oxidation without appetite suppression, stimulant effects, or sympathetic nervous system activation [2].
This mechanism is fundamentally different from conventional fat-loss interventions. Rather than mobilizing fat stores (as with lipolytic agents like AOD 9604) or suppressing appetite (as with GLP-1 agonists), SLU-PP-332 changes which fuel substrate the body preferentially burns — a downstream, utilization-based approach that resembles the metabolic adaptations of trained endurance athletes.
3. Mitochondrial Biogenesis and Cellular Respiration
Through PGC-1α upregulation and activation of oxidative phosphorylation gene networks, SLU-PP-332 promotes the formation of new, functional mitochondria within target cells. In skeletal muscle cell lines (C2C12), treatment increased both mitochondrial density and cellular respiration — two hallmarks of aerobic fitness at the cellular level [1]. This mitochondrial enhancement extends beyond muscle tissue to cardiac, renal, and hepatic cells, as demonstrated in subsequent organ-specific studies [3][4].
4. Non-Hormonal Independence
A critical feature of SLU-PP-332's mechanism is its complete independence from endocrine signaling pathways. The compound does not affect circulating levels of growth hormone, IGF-1, insulin, testosterone, estrogen, or any other endogenous hormone. ERRα, despite containing "estrogen" in its name, is an orphan receptor — it was named for structural similarity to estrogen receptors but does not bind estrogen or participate in estrogen signaling. This non-hormonal mechanism means SLU-PP-332 does not require post-cycle therapy (PCT) and carries no risk of endocrine disruption.
Preclinical Evidence: Exercise Capacity and Skeletal Muscle
The foundational study by Billon et al. (2023) in ACS Chemical Biology established the compound's exercise-mimetic properties through both in vitro and in vivo experiments. In cultured C2C12 skeletal muscle cells, SLU-PP-332 treatment produced significant increases in mitochondrial function and cellular respiration. When administered to mice, the compound produced a measurable increase in Type IIa oxidative muscle fibers — the fiber type associated with sustained aerobic performance and fatigue resistance [1].
Most notably, mice receiving SLU-PP-332 demonstrated enhanced exercise endurance on treadmill testing compared to controls. The researchers further established that blocking ERRα signaling abolished these effects, confirming the receptor-specific mechanism. These data demonstrated the feasibility of targeting ERRα pharmacologically as a strategy for developing exercise mimetics with potential applications in metabolic disease and age-related muscle dysfunction [1].
Metabolic Syndrome and Body Composition
A subsequent study by Billon et al. (2024) in the Journal of Pharmacology and Experimental Therapeutics extended these findings to metabolic disease models. In this 28-day trial, C57BL/6J mice received SLU-PP-332 at 50 mg/kg via intraperitoneal (IP) injection twice daily. The compound was tested in both diet-induced obesity (DIO) and ob/ob genetic obesity mouse models [2].
The metabolic outcomes were notable across multiple parameters. Treated animals demonstrated increased whole-body energy expenditure, enhanced fatty acid oxidation, and a reduced respiratory exchange ratio — indicating a shift in fuel preference from glucose toward lipids. Fat mass was reduced across both obesity models, while insulin sensitivity improved and hepatic steatosis (fatty liver) was markedly reduced [2].
An important observation was that overall body weight did not change significantly despite the reduction in fat mass. This dissociation between total weight and body composition suggests concurrent preservation or improvement of lean tissue — consistent with metabolic reprogramming rather than simple caloric deficit. The body burns more fat as fuel without anorectic effects, behavioral changes, or sympathetic activation [2].
Cardioprotection: Heart Failure and Cardiac Energy Metabolism
The cardiovascular effects of ERR agonism were explored by Xu et al. (2024) in Circulation — one of the highest-impact journals in cardiology. This study examined both SLU-PP-332 and a next-generation analog, SLU-PP-915, in a transverse aortic constriction (TAC) model of pressure overload-induced heart failure in mice [3].
Both compounds produced measurable improvements in cardiac function. Ejection fraction — the percentage of blood pumped out of the left ventricle with each contraction — was preserved or improved in treated animals. Myocardial fibrosis, a hallmark of pathological remodeling in heart failure, was significantly reduced. Survival rates improved compared to untreated controls [3].
The mechanism was traced primarily to ERRγ-mediated enhancement of cardiac fatty acid metabolism and mitochondrial oxidative capacity. The failing heart typically shifts its fuel preference away from fatty acids toward less efficient glucose metabolism — a maladaptive switch that worsens energy deficit and contractile dysfunction. ERR agonism reversed this metabolic remodeling, restoring the heart's preferred energy substrate and improving contractile efficiency. Notably, the compounds did not prevent cardiac hypertrophy itself — they improved cardiac function despite the structural changes associated with pressure overload [3].
These findings position ERR agonists as potential adjunctive therapies for heart failure with reduced ejection fraction (HFrEF), a condition with limited pharmacological options for restoring myocardial energy metabolism.
Renal Protection and Age-Related Decline
Wang et al. (2023) published in the American Journal of Pathology demonstrated that ERR agonism with SLU-PP-332 reversed mitochondrial dysfunction and inflammation in aging kidney tissue. The aging kidney undergoes progressive mitochondrial deterioration, fibrosis, and chronic low-grade inflammation — processes that contribute to the decline in renal function observed in elderly populations [4].
SLU-PP-332 treatment restored mitochondrial architecture and function in aged kidney cells, reduced inflammatory markers, and exhibited anti-fibrotic effects. These findings suggest a role for ERR agonists in counteracting age-related organ decline beyond skeletal muscle and cardiac tissue [4].
A commentary by Nasri (2024) in the Journal of Renal Endocrinology highlighted the dual benefit of metabolic improvement and renal protection, noting that the compound's non-hormonal mechanism makes it particularly attractive for aging populations where hormonal interventions carry elevated risk [6].
Human Myoblast Data: Countering Age-Related Muscle Atrophy
A 2025 pilot study published in Frontiers in Physiology provided the first human-derived tissue data for SLU-PP-332. Researchers cultured myoblasts (muscle precursor cells) isolated from physically inactive human subjects and treated them with SLU-PP-332 in vitro [5].
The results demonstrated several markers of cellular rejuvenation in treated myoblasts:
Reduced cellular damage — Lactate dehydrogenase (LDH) release, an indicator of cell membrane damage, was decreased in treated cultures.
Decreased oxidative stress — Reactive oxygen species (ROS) production was reduced, while glutathione (GSH) levels — a key endogenous antioxidant — were elevated.
Reduced cellular senescence — Senescence-associated β-galactosidase (SA-β-gal) activity, a hallmark of cellular aging, was lower in treated myoblasts.
Enhanced myogenic signaling — SLU-PP-332 upregulated FNDC5 (the precursor of irisin, an exercise-induced myokine), Akt (a central kinase in cell survival signaling), and Bcl-2 (an anti-apoptotic protein). Myotube formation was improved, and myosin heavy chain (MyHC) expression — a marker of mature, functional muscle fibers — was enhanced [5].
These findings suggest that ERR agonism can reverse some of the cellular alterations associated with sedentary aging in human muscle tissue, providing the first translational evidence for this compound class in human-derived cells.
Preclinical Dosing and Pharmacokinetics
All published efficacy studies have used animal models. The standard dosing protocol in mouse studies is 30–50 mg/kg administered via intraperitoneal (IP) injection twice daily, over periods ranging from 12 to 28 days. At 30 mg/kg, plasma concentrations reach approximately 0.2 μM at 6 hours post-injection, with skeletal muscle concentrations approximately threefold higher at 0.6 μM — consistent with the tissue tropism expected for a compound targeting ERRα-rich tissues [1].
Route (preclinical): Intraperitoneal (IP) injection. Oral bioavailability is under investigation in some formulations.
Dose range: 30–50 mg/kg twice daily in mouse models.
Treatment duration: 12–28 days in published studies.
Plasma concentration at 6 hours: Approximately 0.2 μM at 30 mg/kg IP.
Muscle concentration at 6 hours: Approximately 0.6 μM (3× plasma levels) at 30 mg/kg IP.
No human clinical trials have been published. There is no established human dosage, no safety data in humans, and no regulatory approval for clinical use in any jurisdiction. Any dosing protocols circulating in research compound or biohacking communities are extrapolations from animal data and should be interpreted with appropriate caution regarding interspecies pharmacokinetic differences.
Safety Profile: What the Preclinical Data Shows
Across published animal studies, SLU-PP-332 has demonstrated a favorable safety profile within the parameters tested. No liver, kidney, or cardiac toxicity was observed at therapeutic doses over 28-day treatment periods. The compound does not affect endogenous hormone production — it is non-hormonal and does not require post-cycle therapy. No appetite suppression, stimulant effects, or behavioral changes were reported in treated animals [1][2].
However, several critical unknowns remain that must be addressed before clinical translation can be considered:
Uncharacterized parameters include: long-term safety beyond 28 days; drug-drug interactions and hepatic CYP enzyme effects; reproductive and developmental toxicity; carcinogenicity assessment; the safety margin between therapeutic and toxic doses in larger animal species; and pharmacokinetic behavior in human metabolism.
The absence of adverse findings in short-term rodent studies, while encouraging, does not constitute evidence of safety in humans. The history of pharmacology includes numerous examples of compounds with favorable preclinical profiles that produced unexpected toxicity in human trials.
Comparison with Other Metabolic Compounds
SLU-PP-332 vs. AOD-9604
AOD-9604 is a modified fragment of human growth hormone (hGH176-191) that promotes lipolysis — the release of stored fatty acids from adipocytes. Its mechanism acts upstream of energy utilization, liberating fat stores into circulation. SLU-PP-332 operates downstream: it reprograms cellular fuel selection to preferentially oxidize fatty acids. These compounds address different and potentially complementary aspects of fat metabolism — mobilization versus utilization — though no combination studies have been conducted.
SLU-PP-332 vs. Tesamorelin
Tesamorelin (Egrifta®) is an FDA-approved GHRH analog that reduces visceral adipose tissue by stimulating pituitary growth hormone secretion, subsequently elevating IGF-1. It is a hormonal intervention with downstream effects on the GH/IGF-1 axis. SLU-PP-332 operates entirely outside the endocrine system — directly activating metabolic gene programs through nuclear receptor engagement. This non-hormonal mechanism may offer advantages for populations in whom hormonal manipulation is contraindicated, though comparative efficacy data does not exist.
SLU-PP-332 vs. GW501516 (Cardarine)
Both compounds are classified as exercise mimetics, but they act through different nuclear receptor systems. GW501516 is a PPARδ agonist that enhances fatty acid catabolism and endurance. SLU-PP-332 targets the ERR family, activating a broader transcriptional program that includes mitochondrial biogenesis and the acute aerobic exercise response. A notable distinction is that GW501516 development was discontinued due to carcinogenicity concerns in rodent studies, while SLU-PP-332 has not shown such signals in published preclinical data — though the comparative long-term safety profiles remain an open question.
SLU-PP-332 vs. Stimulant-Based Thermogenics
Conventional fat burners (ephedrine, caffeine, clenbuterol) increase energy expenditure through sympathetic nervous system activation — raising heart rate, blood pressure, and core temperature. SLU-PP-332 achieves increased energy expenditure through transcriptional metabolic reprogramming, without sympathomimetic effects. This distinction is clinically relevant for individuals with cardiovascular risk factors, anxiety disorders, or stimulant sensitivity.
Potential Therapeutic Applications
Based on preclinical evidence, SLU-PP-332 and related ERR agonists may have future clinical relevance across several therapeutic domains:
Exercise intolerance and immobility — Individuals unable to exercise due to injury, arthritis, post-surgical recovery, chronic fatigue, or disability could benefit from pharmacological activation of exercise-responsive gene networks. The 2025 Frontiers in Physiology study specifically investigated this application in the context of age-related physical inactivity [5].
Metabolic syndrome — The 2024 Billon study demonstrated improvements in insulin sensitivity, energy expenditure, and hepatic steatosis — three core components of metabolic syndrome — in both genetic and diet-induced obesity models [2].
Heart failure with reduced ejection fraction (HFrEF) — The Xu et al. (2024) Circulation study provides strong preclinical rationale for ERR agonism as an adjunctive therapy in heart failure, particularly in restoring cardiac energy metabolism and reducing fibrosis [3].
Age-related mitochondrial decline — The compound's effects on mitochondrial biogenesis, ROS reduction, and cellular senescence markers suggest potential applications in geriatric medicine and longevity research [4][5].
Chronic kidney disease — The renal-protective effects documented by Wang et al. (2023), including reversal of mitochondrial dysfunction and anti-inflammatory activity, open a therapeutic avenue for slowing age-related kidney function decline [4].
Regulatory Status and Legal Considerations
SLU-PP-332 is currently classified as a research chemical. It is not approved by the FDA, EMA, or any other regulatory agency for human medical use. The compound is available from chemical suppliers for research purposes only and is labeled "not for human consumption."
There is no Good Manufacturing Practice (GMP) certification for SLU-PP-332 production, meaning purity, potency, and contaminant profiles of commercially available material are not guaranteed. Researchers and clinicians should exercise appropriate due diligence regarding source quality and analytical verification.
Of note, a 2026 study by Möller, Krug, and Thevis in Rapid Communications in Mass Spectrometry characterized the in vitro metabolism of both SLU-PP-332 and SLU-PP-915, explicitly evaluating their doping potential [7]. While neither compound currently appears on the World Anti-Doping Agency (WADA) prohibited list, their exercise-mimetic properties make future classification as a prohibited substance a realistic possibility for competitive athletes.
Conclusion
SLU-PP-332 represents a novel approach to metabolic medicine — one that leverages the body's own transcriptional programs for exercise adaptation rather than relying on hormonal manipulation or sympathetic stimulation. The preclinical evidence across skeletal muscle, cardiac, hepatic, and renal systems is compelling, with consistent demonstrations of enhanced fatty acid oxidation, mitochondrial biogenesis, and functional improvement in disease models.
However, scientific rigor demands acknowledgment of this compound's limitations. No human clinical trials have been published. The safety profile, while reassuring in short-term animal studies, remains uncharacterized in humans. The common misidentification of SLU-PP-332 as a "peptide" in online communities reflects a broader pattern of misinformation that can lead to inappropriate expectations, incorrect dosing assumptions, and flawed clinical reasoning.
For researchers and clinicians following this field, the ERR agonist platform — including next-generation compounds like SLU-PP-915 — warrants close attention as it progresses toward potential clinical development. The transition from preclinical promise to validated human therapy will require Phase I safety studies, dose-finding trials, and controlled efficacy studies before SLU-PP-332 or its analogs can be considered for clinical use.
This article is intended for educational and research purposes. SLU-PP-332 is an investigational research compound with no regulatory approval for human use. The information presented here is based on preclinical studies and should not be interpreted as medical advice or as endorsement of off-label use. Consult a qualified healthcare professional before considering any experimental compound.