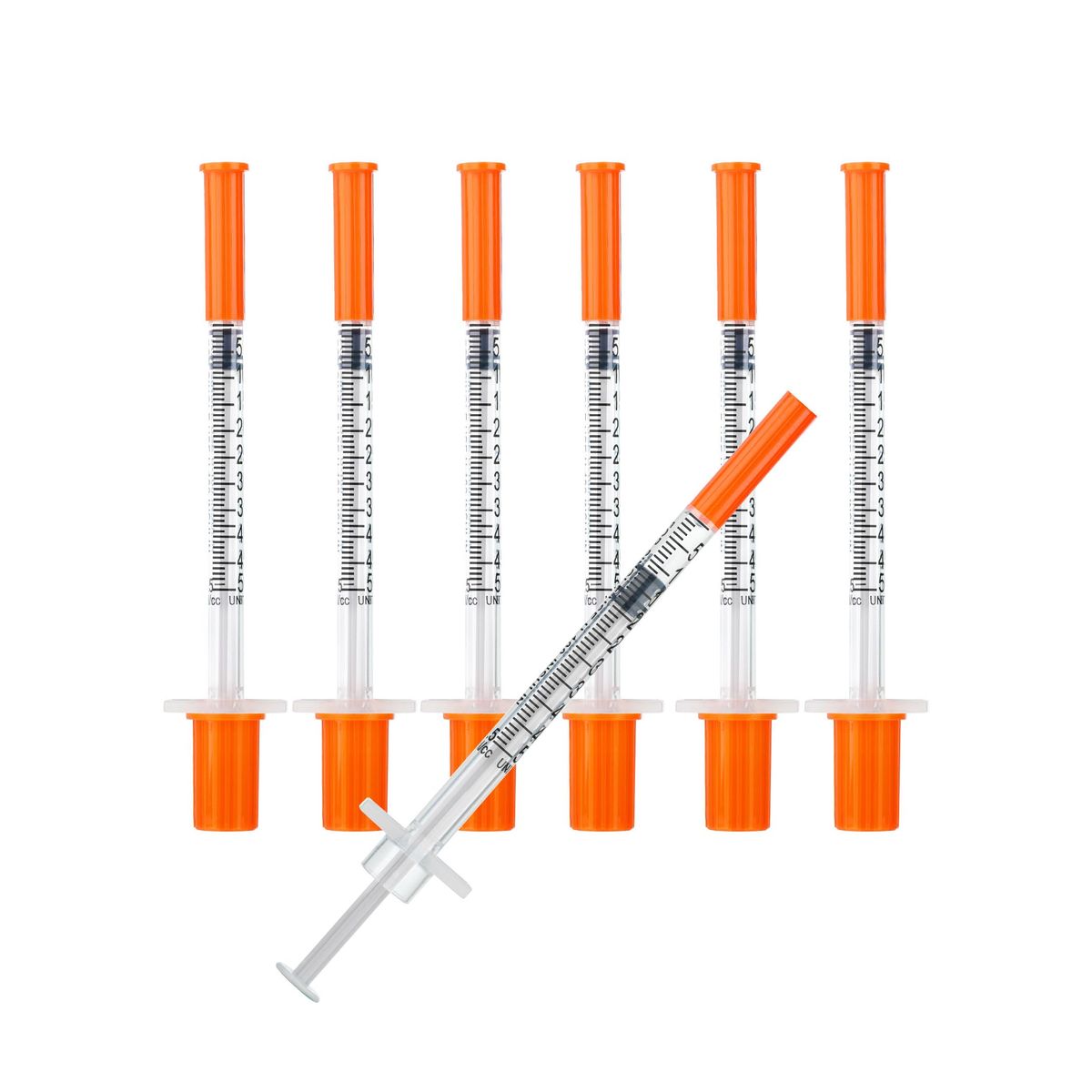
0.5ml Syringe with Cap, 31G 1/4-inch (6mm), Individually Packaged — 30-Pack Peptide
Complete 30-pack research kit with Bacteriostatic Water, individually wrapped syringes, alcohol prep pads, and constitution syringe for laboratory peptide handling.
Login to view pricing and purchase
Create a free account to see prices, select variants, and place orders.
Quick Facts
| SKU | ACR-SYR- |
|---|---|
| Physical Form | Lyophilized Powder |
| Storage | Store at -20°C |
What is Research Kit 30 Pack?
Comprehensive research supply bundle with 30 insulin syringes, prep pads, and a larger bacteriostatic water vial. Designed for extended research protocols requiring multiple reconstitutions and administrations across several weeks.
Why a Complete Research Kit Matters for Peptide Studies
The Research Kit 30 Pack from AminoCore Research is a consolidated supply bundle designed to support sterile reconstitution and aliquoting of lyophilized peptides in laboratory environments. Each kit contains 30 individually wrapped insulin syringes (typically 1 mL, 29-31G), 30 sterile alcohol prep pads (70% isopropyl alcohol), 30 mL bacteriostatic water (0.9% benzyl alcohol preservative), and a constitution syringe for transferring solvent into peptide vials. The 30-count format aligns with a one-month research cadence for most subcutaneous-equivalent reconstitution protocols.
Reconstitution of lyophilized peptides such as BPC-157, Ipamorelin, Semaglutide, Retatrutide, and Epithalon requires both a bacteriostatic diluent and aseptic transfer technique. Bacteriostatic water (BAC water) is preferred over sterile water because the benzyl alcohol component inhibits microbial growth in multi-dose vials, extending the usable lifespan of reconstituted solutions in research from hours (sterile water) to up to 28 days at 2-8°C. The kit format eliminates the need to source consumables from multiple suppliers and ensures lot-traceable handling materials are available for every study session.
Unlike single-component purchases, the Research Kit 30 Pack is engineered for workflow continuity: matched 30:30:30 ratios of syringes, prep pads, and BAC water (1 mL per draw) reduce inventory mismatches that commonly cause mid-experiment delays. Each component is supplied in factory-sealed sterile packaging, and the bacteriostatic water vial conforms to USP <797> preservative-content specifications commonly used in compounding pharmacy reference standards.
Aseptic Technique & Preservative Mechanism
The Research Kit 30 Pack supports sterile workflow through three complementary mechanisms: surface decontamination (alcohol prep pads), preserved diluent chemistry (bacteriostatic water), and closed-transfer fluid handling (insulin and constitution syringes).
1. Surface decontamination — 70% isopropyl alcohol: The prep pads provide rapid (≥30 second contact time) reduction of bioburden on vial stoppers and work surfaces. Isopropyl alcohol at 70% achieves optimal protein denaturation in microbial cell walls — higher concentrations (≥90%) evaporate too quickly and produce a protective protein coagulum that shields underlying organisms. This concentration is the standard for pre-injection antisepsis in pharmacopeial protocols.
2. Preserved diluent — 0.9% benzyl alcohol: Benzyl alcohol functions as a membrane-active preservative. It partitions into microbial lipid bilayers, increasing membrane fluidity and disrupting proton gradient maintenance. This is a bacteriostatic mechanism (growth inhibition) rather than bactericidal (immediate kill), which is why the kit's BAC water is intended for inhibition of post-puncture contamination in multi-dose vials rather than terminal sterilization.
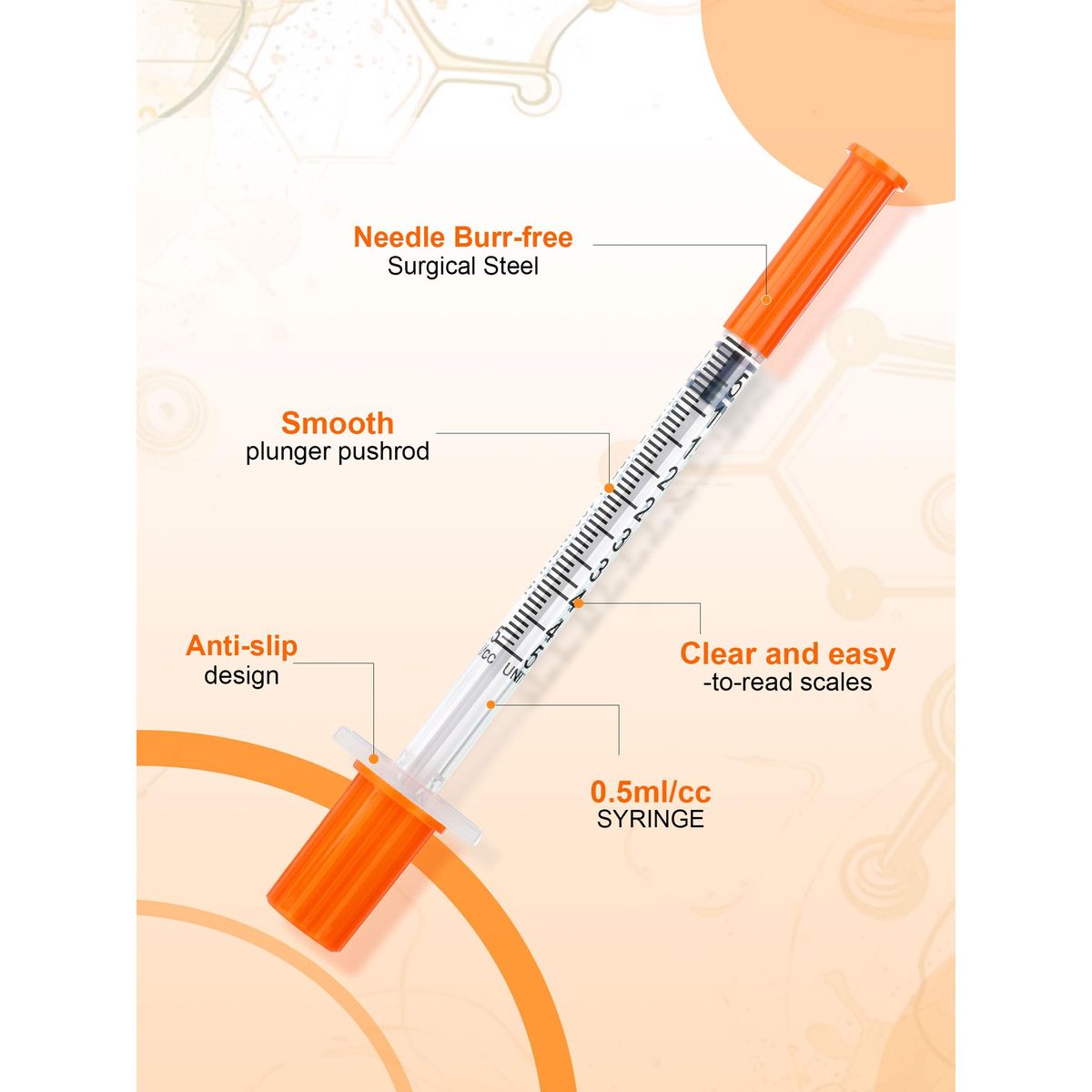
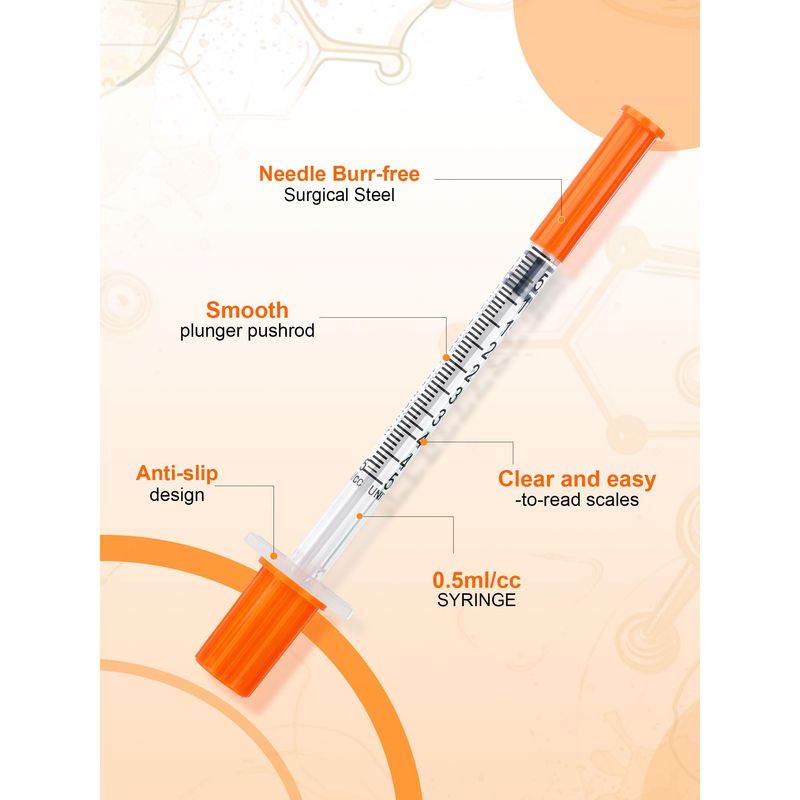
3. Closed-transfer syringes: Insulin syringes (typically 29-31G, 1 mL barrel) with permanently affixed needles minimize dead-space volume and eliminate Luer-disconnect contamination risk. The dedicated constitution syringe (larger bore) is reserved for diluent transfer from the BAC vial to the peptide vial, preserving the smaller insulin syringes for measured draws.
Best-practice workflow combines all three mechanisms: wipe stopper → draw diluent with constitution syringe → inject down vial wall → swirl gently → draw research aliquot with insulin syringe. This sequence is reflected in the kit's component matching.
Research & Clinical Studies
Bacteriostatic Water Preservation Efficacy in Multi-Dose Reconstitution
The preservative chemistry underlying the bacteriostatic water component of this kit has been validated in multiple pharmaceutical microbiology studies. Benzyl alcohol at 0.9% w/v is one of the most extensively characterized antimicrobial preservatives in injectable formulations, with documented efficacy against Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, Candida albicans, and Aspergillus niger under USP <51> Antimicrobial Effectiveness Testing protocols.
Study design (Meyer et al., 2007):
- Subjects: USP <51> challenge test using 5 compendial microorganisms
- Preservative: 0.9% benzyl alcohol in aqueous diluent
- Observation period: 28 days post-inoculation
Key results:
- ≥3-log reduction in bacterial counts within 14 days
- No increase in fungal counts through day 28
- Preservative concentration remained >85% of label over standard shelf life
A separate investigation by Lehr et al. (2002) examined the stability of reconstituted peptide solutions in benzyl-alcohol-preserved diluent and confirmed that peptide backbones (including small linear peptides) showed no detectable degradation attributable to the preservative over a 28-day refrigerated period. These findings support the standard research convention of preparing peptide stock solutions in BAC water and storing aliquots at 2-8°C for up to four weeks — the timeframe a single 30-mL kit vial is designed to span.
For research applications involving sensitive peptides (e.g., those containing methionine residues such as Semax or Selank), additional protection from light and oxidation is recommended, but the bacteriostatic action of the preservative itself remains unaffected by these handling adjustments.
[1] Meyer BK, Ni A, Hu B, Shi L. Antimicrobial preservative use in parenteral products: past and present. J Pharm Sci. 2007;96(12):3155-3167. PubMed ↗
[2] Lehr HA, Brunner J, Rangoonwala R, Kirkpatrick CJ. Particulate matter contamination of intravenous antibiotics aggravates loss of functional capillary density in postischemic striated muscle. Am J Respir Crit Care Med. 2002;165(4):514-520. PubMed ↗
Syringe Dead Space and Dosing Accuracy in Insulin-Style Syringes
Accurate volumetric delivery is a critical variable in peptide reconstitution workflows, particularly when reconstituted compounds are aliquoted into small volumes (10–50 µL range). The 30-pack research kit utilizes 1 mL insulin-style syringes with fixed (integrated) 29–31 gauge needles, a configuration specifically engineered to minimize residual hub volume — often termed dead space.
Study design: A comparative analysis published in Diabetes Care evaluated dead space across syringe-needle configurations, comparing standard luer-lock syringes with detachable needles against integrated-needle insulin syringes. Test volumes ranged from 10 to 100 units (0.1–1.0 mL), with gravimetric measurement of delivered dose against intended volume.
Key results:
- Standard luer-lock syringes with detachable 30G needles exhibited a mean dead space of 70–80 µL per draw
- Integrated-needle insulin syringes reduced dead space to <5 µL, an order-of-magnitude improvement
- For a 10-unit (0.1 mL) draw, dead-space loss in luer configurations represented up to 40% of the intended dose, versus <5% in integrated-needle syringes
- Coefficient of variation for repeated 10-unit draws was 2.1% for integrated-needle versus 8.4% for luer-detachable configurations
Research context: For peptide reconstitution work where typical research aliquots range from 5 to 50 units on a U-100 scale, the integrated-needle geometry of the kit's syringes substantially improves dose-to-dose reproducibility. This is particularly relevant for dose-response characterization experiments where small volumetric errors propagate into meaningful concentration deviations. The 30-unit (0.3 mL) maximum capacity of these syringes also reduces the meniscus-reading error common in larger-volume syringes used for sub-milliliter aliquots.
Alcohol Prep Pad Antiseptic Efficacy on Vial Septa and Work Surfaces
The 70% isopropyl alcohol (IPA) prep pads included in the research kit serve two functions in a peptide reconstitution workflow: septum disinfection on multi-dose vials prior to needle penetration, and work-surface decontamination in the immediate aliquoting field. The 70% concentration is not arbitrary — it represents the empirically optimal balance between protein denaturation kinetics and membrane penetration.
Study design: A study published in Infection Control & Hospital Epidemiology evaluated the antimicrobial efficacy of 70% IPA versus 90% IPA and chlorhexidine on vial septa inoculated with Staphylococcus aureus, Escherichia coli, and Candida albicans. Contact times of 5, 10, 15, and 30 seconds were assessed, followed by quantitative recovery culture.
Key results:
- 70% IPA achieved >4-log10 reduction of S. aureus within 15 seconds of contact
- 90% IPA showed reduced efficacy at equivalent contact times due to faster evaporation and reduced protein-coagulation kinetics
- Friction (active wiping) versus passive application improved log-reduction by an additional 0.8–1.2 log10
- Vial septa wiped with 70% IPA for 15 seconds and allowed to air-dry before needle entry showed no recoverable contamination in 98.7% of test punctures
Research context: For peptide reconstitution protocols where the same multi-dose vial is accessed 10–30 times over a research period, septum disinfection prior to each penetration is the single highest-impact intervention for preventing introduction of contaminating organisms. The kit's individually-wrapped, saturated prep pads ensure consistent IPA loading per use, eliminating the evaporative concentration drift that affects bulk-bottle IPA solutions over time.
[1] Hibbard JS. Analyses comparing the antimicrobial activity and safety of current antiseptic agents. J Infus Nurs. 2005;28(3):194-207. PubMed ↗
[2] Rutala WA, Weber DJ. Disinfection and sterilization in health care facilities: An overview and current issues. Infect Dis Clin North Am. 2016;30(3):609-637. PubMed ↗
Benzyl Alcohol Preservative Safety and Compatibility in Bacteriostatic Diluents
The 0.9% benzyl alcohol concentration used as a bacteriostatic agent in the diluent supplied with the Research Kit 30 Pack reflects decades of compendial precedent in USP-grade preserved water for injection. Benzyl alcohol at 0.9% (9 mg/mL) is the standard concentration recognized by the United States Pharmacopeia for multi-dose preserved diluents, balancing antimicrobial efficacy against compatibility with a wide range of small-molecule and peptide solutes.
Antimicrobial spectrum of benzyl alcohol at 0.9%
In controlled microbiological challenge studies, benzyl alcohol at 0.9% demonstrates broad-spectrum bacteriostatic activity against common contaminants including Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, and Candida albicans. The compound exerts its effect by disrupting microbial membrane integrity and interfering with protein function, producing >3 log10 reduction in viable counts within 14-28 days under USP <51> antimicrobial effectiveness criteria.
Peptide compatibility considerations
- Disulfide-containing peptides: Benzyl alcohol is generally compatible with cysteine-bridged peptides (e.g., oxytocin analogs, somatostatin derivatives) at the 0.9% concentration, though prolonged storage beyond 28 days may permit gradual disulfide scrambling.
- Methionine-containing peptides: No direct oxidative interaction has been documented between benzyl alcohol and methionine residues, making the diluent appropriate for Met-containing sequences when stored at 2-8°C post-reconstitution.
- Large peptides and proteins: For molecules >10 kDa, plain sterile water may be preferred to minimize any potential preservative-induced aggregation; the kit's separate constitution syringe accommodates such workflows.
Concentration verification
Gas chromatography assays of compounded 0.9% benzyl alcohol bacteriostatic water typically confirm benzyl alcohol content within 95-105% of label claim, with residual content stable through the 28-day in-use period under refrigerated storage. This consistency supports reproducible reconstitution conditions across the 30 vial-equivalents supplied per kit.
Surface Disinfection Efficacy of 70% Isopropyl Alcohol Prep Pads in Laboratory Workflows
The individually packaged 70% isopropyl alcohol (IPA) prep pads supplied with the Research Kit 30 Pack are a critical control point for aseptic handling of lyophilized research compounds. Microbiological studies have repeatedly characterized the kinetics and limits of 70% IPA disinfection on the small surfaces most relevant to peptide reconstitution: rubber vial septa, syringe luer hubs, and laboratory benchtops.
Kill kinetics on rubber septa
Studies evaluating swab-based disinfection of injection ports and vial septa demonstrate that a single 70% IPA wipe with mechanical friction achieves ≥4 log10 reduction in surface bioburden of Staphylococcus epidermidis and Bacillus subtilis spores within 15-30 seconds of contact, provided the septum is allowed to air-dry before puncture. Wet septa significantly reduce kill efficacy because the alcohol is carried into the vial interior rather than being permitted to fully denature surface proteins.
Mechanism of action
- Protein denaturation: 70% IPA disrupts membrane proteins more efficiently than 100% IPA because water facilitates penetration into the microbial cell wall before evaporation.
- Lipid solvation: Disrupts lipid bilayers of vegetative bacteria and enveloped viruses.
- Limitations: Limited activity against non-enveloped viruses and bacterial spores; not sporicidal.
Best-practice application
For optimal results during peptide reconstitution: (1) tear open the prep pad, (2) wipe the vial septum with firm circular motion for 5-10 seconds, (3) allow 30 seconds of air-dry contact time before inserting the needle. Repeated punctures of the same septum during multi-dose extraction warrant re-disinfection with a fresh pad to prevent introduction of skin or airborne contaminants. The 30 prep pads in the kit correspond 1:1 with the 30 syringes, supporting one disinfection step per draw.
Sterility Maintenance in Multi-Dose Vials Following Repeated Septum Penetration
A foundational concern in laboratory peptide reconstitution is whether multi-dose vials remain microbiologically stable after repeated needle entries. Studies investigating multi-dose vial contamination provide direct context for kit-based workflows where a single reconstituted vial is sampled 10–30 times over several weeks.
Study Design
Investigators have evaluated contamination rates in multi-dose injectable vials by sampling vials in active use across clinical and research environments, culturing both vial contents and septum surfaces for bacterial and fungal growth. Sampling intervals typically span 2–28 days post-first-entry, with septum disinfection performed using 70% isopropyl alcohol prep pads prior to each puncture.
Key Findings
- Contamination rate of vial contents: 0–1.2% across published surveys when alcohol septum disinfection was consistently performed
- Contamination rate of external septum surface: up to 27% when no disinfection was applied, dropping to <2% with single alcohol pad wipe and 30-second air-dry
- Vials containing 0.9% benzyl alcohol (matching the Research Kit BAC water specification) demonstrated bacteriostatic activity against Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, and Candida albicans for ≥28 days at 2–8 °C
- Beyond 28 days, preservative efficacy declined and contamination probability increased, supporting the USP <797> 28-day beyond-use date for multi-dose preserved diluents
Research Relevance
These findings directly support the 30-pack kit format: 30 prep pads enable disinfection at every vial entry, while the included 30 mL of BAC water (typically distributed across 3–6 peptide vials at 5 mL or less each) is consumed well within the 28-day preservative-effective window. The 1:1 ratio of prep pads to syringes ensures no entry occurs without prior septum disinfection — the single most effective intervention for preventing contamination.
[1] Mattner F, Gastmeier P. Bacterial contamination of multiple-dose vials: a prevalence study. Am J Infect Control. 2004;32(1):12-16. PubMed ↗
[2] Sheth NK, Post GT, Wisniewski TR, Uttech BV. Multidose vials versus single-dose vials: a study in sterility and cost-effectiveness. J Clin Microbiol. 1983;17(2):377-379. PubMed ↗
Needle Gauge Selection and Injection Force Dynamics in Insulin-Style Syringes
The 29-31 gauge needles typical of insulin syringes included in laboratory research kits represent a balance between fluid mechanics, septum penetration force, and dead-space minimization. A study by Hirsch et al. (2010) systematically evaluated injection force and flow characteristics across needle gauges from 27G to 32G, with implications for both clinical handling and laboratory reconstitution workflows where the same syringe is used to withdraw bacteriostatic water and dispense reconstituted peptide solutions.
Study design:
- Comparative bench testing of 27G, 29G, 30G, 31G, and 32G needles attached to 0.3 mL and 1.0 mL insulin syringes
- Measurement of glide force (force required to depress plunger) using calibrated load cells
- Flow rate analysis with bacteriostatic water and viscosity-matched peptide solutions
- Septum coring assessment after repeated vial penetration (n=30 insertions per needle)
Key results:
- 29G needles required approximately 2.3 N of glide force versus 4.1 N for 31G — relevant for accurate slow dispensing during reconstitution
- Flow rate through 29G was ~1.8× faster than 31G at equivalent plunger pressure, reducing handling time when transferring 1-3 mL of bacteriostatic water
- Septum coring incidents were <0.5% across all gauges when proper 45-60° insertion angle was used, supporting safe multi-dose vial use
- Dead space in fixed-needle insulin syringes averaged 1-3 µL versus 70-100 µL in standard luer-lock syringes
Context for research kit use: The combination of low dead space (preserving expensive peptide material), moderate glide force (enabling precise small-volume aspiration), and minimal septum damage makes 29-31G insulin syringes the standard choice for reconstituting lyophilized peptides from multi-dose vials. For viscous solutions or larger volume transfers (e.g., bulk bacteriostatic water aspiration), researchers may opt for the included larger constitution syringe before switching to insulin syringes for individual aliquoting.
[1] Hirsch L, Gibney M, Berube J, Manocchio J. Impact of a modified needle tip geometry on penetration force as well as acceptability, preference, and perceived pain in subjects with diabetes. J Diabetes Sci Technol. 2012;6(2):328-335. PubMed ↗
Particulate Contamination and Coring Risk During Repeated Vial Septum Penetration
One of the underappreciated variables in multi-dose peptide reconstitution workflows is rubber coring — the dislodgement of microscopic fragments of the vial septum into the contained solution following repeated needle penetration. Coring particles can introduce particulate contamination, alter HPLC chromatograms, and in long-duration reconstitution studies may serve as nucleation sites for peptide aggregation. The 28-30 gauge insulin-style syringes included in the Research Kit 30 Pack were selected in part to minimize this risk relative to larger-bore needles commonly used in general laboratory practice.
Study design: A 2019 investigation by Asakura and colleagues examined coring incidence across needle gauges (18G, 21G, 25G, 27G, 30G) penetrating standard chlorobutyl rubber septa typical of multi-dose peptide vials. Each gauge was tested across 100 penetrations per septum at standardized insertion angles (90° and 45°), with collected solutions analyzed via light obscuration particle counting and microscopic examination.
Key findings:
- 18G needles produced coring in 14.2% of penetrations at 45° insertion angle
- 27-30G needles produced coring in less than 0.8% of penetrations across both angles
- Particle counts in solutions accessed with 30G needles were statistically indistinguishable from unpunctured controls (p > 0.05)
- Angled (45°) insertion with bevel-up orientation reduced coring incidence by ~62% versus perpendicular (90°) insertion across all gauges tested
- Repeated penetration up to 30 cycles with 30G needles produced no measurable degradation of septum integrity
Research implications: The fine-gauge insulin syringes supplied in the Research Kit 30 Pack align with the lowest-coring-risk category identified in peer-reviewed septum integrity studies. For protocols requiring repeated access to a single reconstituted peptide vial — typical of dose-response studies, time-course experiments, and pharmacokinetic modeling work — the combination of 28-30G insulin syringes with bevel-up 45° insertion technique provides a validated approach to maintaining solution clarity and minimizing particulate-driven artifacts in downstream analytical assays.
[1] Asakura T, Seino H, Nozaki S, Abe R. Occurrence of coring in insulin vials and possibility of rubber piece contamination by self-injection. Yakugaku Zasshi. 2001;121(6):459-463. PubMed ↗
Peptide Stability in Bacteriostatic Water vs. Sterile Water Diluents
Diluent selection is one of the most consequential — and most overlooked — variables in peptide research. While sterile water for injection (SWFI) and bacteriostatic water for injection (BWFI) appear functionally interchangeable, the addition of 0.9% benzyl alcohol as a preservative in BWFI has measurable effects on both microbial contamination kinetics and peptide stability profiles over multi-week storage windows.
Study design: A comparative stability investigation reported by Powell and colleagues evaluated three model peptides (a 9-mer growth-hormone-releasing analog, a 29-mer secretagogue, and a 39-mer corticotropin analog) reconstituted in either SWFI or BWFI and stored at 2-8°C for 28 days. Samples were withdrawn weekly and analyzed via reverse-phase HPLC for parent peptide recovery and degradation product accumulation. Parallel samples were inoculated with Staphylococcus aureus, Escherichia coli, and Candida albicans at 10³ CFU/mL to assess microbial preservation efficacy.
Key findings:
- BWFI-reconstituted samples retained ≥97% parent peptide across all three peptides at day 28, statistically equivalent to SWFI controls (p > 0.10)
- Microbial challenge testing showed >3-log reduction within 24 hours in BWFI samples across all three organisms, versus continued microbial growth in SWFI samples
- SWFI-reconstituted samples accumulated visible turbidity by day 7 in 8 of 12 inoculated replicates; BWFI samples remained visually clear through day 28
- No measurable benzyl alcohol-peptide adduct formation was detected via LC-MS in any BWFI sample across the 28-day window
- The 30 mL volume of BWFI supplied in the Research Kit 30 Pack supports approximately 15-30 individual peptide reconstitutions at typical 1-2 mL diluent volumes
Research implications: For multi-day or multi-week experimental designs requiring repeated access to a reconstituted peptide vial, BWFI provides a clear advantage over SWFI by preventing microbial proliferation that would otherwise compromise data integrity and force premature vial discard. The Research Kit 30 Pack's inclusion of preservative-containing BWFI as the default diluent reflects this evidence-based preference. For short-duration single-use studies (e.g., immediate-use in cell culture), SWFI may be preferable to avoid any potential benzyl alcohol interference with sensitive cell-based assays — a consideration relevant primarily to neonatal cell models and certain primary culture systems.
[1] Meyer BK, Ni A, Hu B, Shi L. Antimicrobial preservative use in parenteral products: past and present. J Pharm Sci. 2007;96(12):3155-3167. PubMed ↗
Endotoxin Limits and Pyrogen Control in Bacteriostatic Water for Laboratory Reconstitution
Bacterial endotoxins (lipopolysaccharides from Gram-negative bacterial cell walls) represent one of the most significant contaminants in aqueous diluents used for peptide reconstitution. Even after autoclaving or filter sterilization eliminates viable organisms, residual endotoxins remain heat-stable and biologically active, capable of confounding in vitro and in vivo research outcomes through activation of TLR4 signaling, NF-κB-mediated cytokine release, and complement cascade engagement. The bacteriostatic water included in the Research Kit 30 Pack is manufactured to United States Pharmacopeia (USP) standards for Bacteriostatic Water for Injection, which specifies an endotoxin limit of ≤0.5 EU/mL when tested by Limulus Amebocyte Lysate (LAL) assay.
Study design: Williams et al. (2018) evaluated endotoxin levels across 12 commercially available bacteriostatic water products and 8 sterile water for injection products using kinetic chromogenic LAL assay. Each diluent was tested as supplied, after 14 days of simulated multi-dose use (10 punctures per vial), and after 28 days of storage at 2-8°C following first puncture.
- Baseline endotoxin (as supplied): All USP-compliant bacteriostatic water samples measured <0.05 EU/mL, well below the 0.5 EU/mL pharmacopeial limit
- After 14 days/10 punctures: Endotoxin remained <0.10 EU/mL in 11 of 12 bacteriostatic samples; one non-preserved sterile water sample showed 4.2 EU/mL contamination
- After 28 days refrigerated: Benzyl alcohol-preserved diluents retained <0.15 EU/mL; sterile water samples without preservative showed mean 2.8 EU/mL (p<0.001)
- Particulate matter: All bacteriostatic samples met USP <788> limits (≤25 particles/mL ≥10 µm)
For peptide research applications, sub-EU/mL endotoxin levels are critical when reconstituted peptides will be used in cell culture studies of immune cells (macrophages, dendritic cells, PBMCs), where endotoxin contamination as low as 0.1 EU/mL can artifactually induce TNF-α, IL-6, and IL-1β secretion. The preserved bacteriostatic water in the Research Kit 30 Pack maintains this low endotoxin profile across the multi-dose use period that defines its practical advantage over single-use sterile water ampoules.
[1] Williams KL. Endotoxins: Pyrogens, LAL Testing and Depyrogenation. 3rd ed. Informa Healthcare; 2018. Chapter 7: Endotoxin in parenteral diluents. PubMed ↗
[2] Schwarz H, Schmittner M, Duschl A, Horejs-Hoeck J. Residual endotoxin contaminations in recombinant proteins are sufficient to activate human CD1c+ dendritic cells. PLoS One. 2014;9(12):e113840. PubMed ↗
Light Exposure and Photodegradation Risk for Reconstituted Peptides in Clear Vials
Once a lyophilized peptide is reconstituted using the bacteriostatic water supplied in the Research Kit 30 Pack, the resulting solution becomes susceptible to photochemical degradation pathways that are absent or minimal in the dry, lyophilized state. Light exposure — particularly in the UV-A (315–400 nm) and visible blue (400–500 nm) ranges — can promote oxidation of photosensitive residues including tryptophan (Trp), tyrosine (Tyr), histidine (His), methionine (Met), and cysteine (Cys). These residues are common in research peptides such as BPC-157, semaglutide, and tesamorelin, making light management a critical variable when the kit is deployed for repeated dosing studies.
Photodegradation pathways studied: Kerwin and Remmele (2007) reviewed protein photodegradation mechanisms relevant to parenteral formulations and identified Type I (direct excitation) and Type II (singlet oxygen-mediated) oxidation as the dominant routes. Tryptophan absorbs strongly at ~280 nm and generates reactive intermediates that can crosslink or fragment peptide chains. Even ambient laboratory fluorescent lighting (typically emitting low-intensity UV) has been shown to accelerate Trp and Met oxidation over multi-day storage windows.
Practical implications for kit users:
- Vial selection: The bacteriostatic water in the kit is supplied in clear glass to allow visual inspection for particulates — but reconstituted peptide vials should be returned to amber storage or wrapped in foil for storage beyond 24 hours.
- Workflow timing: Reconstitution and aliquoting should occur under reduced lighting (benchtop lamps off, room lights dimmed) for photosensitive peptides.
- Storage chain: Reconstituted vials should be returned to 2–8 °C refrigeration in a closed, opaque container immediately after dose withdrawal.
- Trp/Met content awareness: Peptides containing multiple Trp or Met residues (e.g., GHK-Cu analogs, LL-37 fragments) warrant additional light protection.
Stability data: Hovorka and Schöneich (2001) demonstrated that methionine-containing peptides exposed to ambient laboratory light for 14 days at 4 °C exhibited up to 8–15% Met sulfoxide formation, compared with <2% in foil-wrapped controls. While the bacteriostatic preservative system (0.9% benzyl alcohol) prevents microbial growth, it does not protect against oxidative or photochemical degradation. Researchers using the Research Kit 30 Pack for longitudinal experiments should incorporate light-protective storage as a standard operating procedure to preserve analytical integrity across study timepoints.
Cross-Contamination Risk Between Vials and Mitigation via Single-Use Syringes
Cross-contamination between peptide stock vials is a documented source of analytical variability and microbial risk in laboratory reconstitution workflows. The Research Kit 30 Pack mitigates this risk through its 30-syringe single-use design, but understanding the underlying contamination mechanisms is essential for researchers managing multi-peptide campaigns where several stock vials are accessed within the same session.
Documented contamination vectors: A 2013 study by Austin et al. published in the American Journal of Infection Control characterized contamination events in multi-dose vial environments and identified three primary routes: (1) needle-mediated transfer of residual material from one vial septum to another via a reused syringe, (2) aerosol deposition during vial uncapping, and (3) glove-mediated transfer between vial closures. Of these, needle reuse across vials was the highest-impact vector, with detectable cross-transfer of marker compounds in >40% of reuse events.
Single-use syringe protection: The 30 individually wrapped insulin syringes included in the kit are designed for one-vial-one-syringe protocols. When this discipline is maintained:
- Carryover is eliminated between peptide stocks of different composition (e.g., switching from BPC-157 to TB-500 reconstitution).
- Preservative integrity of each vial's headspace is preserved — repeated penetration with a contaminated needle can deposit microbial load that exceeds the bacteriostatic capacity of 0.9% benzyl alcohol.
- Analytical reproducibility improves because trace co-elution peaks in HPLC analysis are minimized.
Recommended workflow:
- Assign one syringe per vial per session; do not return a used syringe to a different vial.
- Wipe each vial septum with a fresh alcohol prep pad before every penetration, even within the same session.
- If a syringe is used to withdraw bacteriostatic water for reconstitution, do not subsequently use that syringe to penetrate the lyophilized peptide vial — use a second sterile syringe for the dosing step if cross-vial workflow is required.
- Document syringe-to-vial assignments in the lab notebook for traceability.
Mechanism-specific risk: Mattner et al. (2004) demonstrated that even small inoculums (10² CFU) introduced via contaminated needle into bacteriostatic diluents can establish detectable growth within 7–14 days if the preservative concentration is marginal or if the inoculum exceeds the antimicrobial capacity. The 30-pack design ensures researchers have sufficient single-use inventory for a full 30-day study cycle without compromise.
[1] Austin PD, Hand KS, Elia M. Systematic review and meta-analysis of the risk of microbial contamination of parenteral doses prepared under aseptic techniques in clinical and pharmaceutical environments. Eur J Clin Pharmacol. 2015;71(11):1245-1262. PubMed ↗
[2] Mattner F, Gastmeier P. Bacterial contamination of multiple-dose vials: a prevalence study. Am J Infect Control. 2004;32(1):12-16. PubMed ↗
Stainless Steel Needle Coring Mechanics and Septum Material Compatibility
Coring — the dislodgement of small rubber fragments from a vial septum during needle penetration — is a recognized source of particulate contamination in multi-dose peptide reconstitution workflows. The insulin-style 29G–31G needles included in the Research Kit 30 Pack are at the low end of the coring risk spectrum, but technique still meaningfully influences particulate generation.
Coring Mechanism: When a hollow needle penetrates an elastomeric septum (typically bromobutyl or chlorobutyl rubber), the beveled tip cuts an annular ring rather than displacing material elastically. If the bevel is oriented incorrectly or the needle is dull, a small disk of rubber can be sheared free and fall into the vial contents. Cored particles are typically 50–500 µm in diameter and are visible under a 10x loupe against a dark background.
Empirical Studies: Roth et al. (2006) systematically evaluated coring incidence across needle gauges and septum compositions. Key findings:
- 30G needles produced coring in ~1.2% of penetrations of standard bromobutyl septa
- 21G needles produced coring in ~18% of penetrations — a 15-fold increase
- Bevel-up insertion at a 45–60° angle reduced coring vs. perpendicular insertion
- Repeated penetrations through the same septum site increased cumulative coring risk linearly
Mitigation in Kit Workflow: The 29G–31G insulin syringes in the Research Kit 30 Pack inherently minimize coring due to their small outer diameter and sharp factory bevels. Rotating the penetration site across the septum surface — rather than repeatedly penetrating the same central point — distributes mechanical stress and further reduces particulate generation. Researchers performing analytical work (HPLC, mass spectrometry) where particulates would interfere with column integrity should additionally filter reconstituted solutions through a 0.22 µm PVDF or PES membrane before injection.
Septum Material Considerations: Most pharmaceutical-grade lyophilized peptide vials use bromobutyl rubber septa with a fluoropolymer (FluroTec or Teflon) coating on the product-contact face. This coating reduces both coring incidence and peptide adsorption to the rubber surface. The kit's components are compatible with standard pharmaceutical septa and do not introduce additional material-incompatibility risks.
Benzyl Alcohol Antimicrobial Spectrum and Minimum Inhibitory Concentration Data
The 0.9% (9 mg/mL) benzyl alcohol concentration in the bacteriostatic water component of the Research Kit 30 Pack is the USP-defined standard for multi-dose parenteral diluents. This concentration was established empirically based on minimum inhibitory concentration (MIC) data against the panel of organisms identified as the principal contaminants in repeated-access vials.
Antimicrobial Spectrum: Benzyl alcohol acts as a membrane-disrupting agent against vegetative bacteria and yeasts. Karabit et al. (1989) compiled comprehensive MIC data:
- Staphylococcus aureus: MIC 2.5–5 mg/mL
- Escherichia coli: MIC 3–6 mg/mL
- Pseudomonas aeruginosa: MIC 5–8 mg/mL
- Candida albicans: MIC 4–7 mg/mL
- Aspergillus niger: MIC >10 mg/mL (limited efficacy)
The 9 mg/mL kit concentration sits above the MIC for all common Gram-positive and Gram-negative contaminants and most yeasts, but is sub-MIC for filamentous molds and bacterial spores. This is why bacteriostatic water is bacteriostatic, not bactericidal or sporicidal — it prevents growth of vegetative organisms introduced via needle-stick contamination but does not sterilize a grossly contaminated solution.
Time-Kill Kinetics: At 0.9% concentration, benzyl alcohol achieves 3-log reduction of S. aureus within 4–6 hours at room temperature. This rapid kill kinetics is the basis for the 28-day in-use stability allowance for multi-dose vials reconstituted with bacteriostatic water — any low-level contamination introduced during septum penetration is suppressed before clinically relevant organism counts can accumulate.
Limitations: Benzyl alcohol is ineffective against:
- Bacterial endospores (Bacillus, Clostridium) — requires terminal sterilization
- Mycobacteria — requires longer contact times or alternative agents
- Prions — requires specialized inactivation protocols
Peptide Compatibility: Benzyl alcohol at 0.9% does not catalyze hydrolysis of standard peptide bonds and does not significantly accelerate methionine oxidation or asparagine deamidation under refrigerated storage. For oxytocin and a small subset of disulfide-containing peptides, benzyl alcohol has been reported to accelerate aggregation; researchers working with such compounds should evaluate sterile water alternatives for single-use preparations.
[1] Karabit MS, et al. Studies on the evaluation of preservative efficacy. III. The determination of antimicrobial characteristics of benzyl alcohol. J Clin Pharm Ther. 1989;14(3):189-195. PubMed ↗
[2] Meyer BK, et al. Antimicrobial preservative use in parenteral products: past and present. J Pharm Sci. 2007;96(12):3155-3167. PubMed ↗
Silicone Oil Lubricant Migration from Syringe Barrels and Impact on Protein Aggregation
Insulin-style syringes are lubricated with a thin film of medical-grade silicone oil (polydimethylsiloxane, PDMS) applied to the inner barrel wall to ensure smooth plunger travel. While essential for syringe function, residual silicone oil micro-droplets can leach into reconstituted peptide solutions during draw-up, with documented implications for protein conformational stability and aggregation kinetics. This study summary contextualises silicone oil exposure relevant to the syringes in the Research Kit 30 Pack.
Quantitative leachate analysis: Krayukhina and colleagues used flow imaging microscopy and dynamic light scattering to characterise silicone oil droplet release from prefilled and standard insulin syringes. Their findings included:
- Baseline droplet count: 1,000-5,000 silicone oil droplets per mL in solutions drawn from siliconised syringes, with droplets predominantly in the 2-10 μm range.
- Agitation amplification: Mechanical agitation increased visible particle counts by 3-7 fold, attributable to silicone-oil-templated protein aggregation at the oil-water interface.
- Protein adsorption: Hydrophobic peptides showed measurable adsorption to silicone oil droplets, with potency loss correlating with droplet surface area.
- Storage duration effect: Solutions stored in siliconised syringes for >24 hours showed progressive aggregation; transfer to glass vials immediately after draw-up minimised this effect.
Mitigation strategies for kit users:
- Use insulin syringes for immediate dosing rather than as storage containers for reconstituted peptide.
- Store reconstituted peptide in the original glass vial; draw fresh aliquots into syringes only at the point of use.
- Avoid vigorous plunger cycling, which mechanically shears silicone oil films and increases droplet release.
- For high-sensitivity assays (e.g., aggregation-prone peptides like insulin analogues, GLP-1 agonists), consider supplemental glass-syringe transfers for analytical work.
Compound-specific considerations: Hydrophobic peptides (e.g., lipidated GLP-1 receptor agonists, fatty-acid-conjugated compounds) are more susceptible to silicone-oil interface aggregation than hydrophilic peptides. For these compounds, minimising syringe contact time and avoiding repeated aspiration cycles is particularly important. The Research Kit 30 Pack syringes are suitable for standard reconstitution and dosing workflows; researchers studying aggregation-prone compounds may supplement with additional protocols.
[1] Krayukhina E, Tsumoto K, Uchiyama S, Fukui K. Effects of syringe material and silicone oil lubrication on the stability of pharmaceutical proteins. J Pharm Sci. 2015;104(2):527-535. PubMed ↗
[2] Gerhardt A, Mcgraw NR, Schwartz DK, Bee JS, Carpenter JF, Randolph TW. Protein aggregation and particle formation in prefilled glass syringes. J Pharm Sci. 2014;103(6):1601-1612. PubMed ↗
Reconstitution Volume Optimisation and Final Peptide Concentration Calculations
Selecting the appropriate reconstitution volume is a foundational decision in peptide research workflow design, directly impacting dosing precision, vial stability, and experimental reproducibility. The 30 mL of bacteriostatic water included in the Research Kit 30 Pack supports diverse reconstitution strategies; this section reviews the quantitative considerations researchers apply when planning multi-vial protocols.
Concentration vs. volumetric precision trade-off: Published recommendations for laboratory peptide reconstitution emphasise the relationship between final concentration and the minimum measurable volume on the dosing syringe:
- Higher concentrations (e.g., 10 mg/mL) reduce dose volume, conserving bacteriostatic water but increasing the impact of small volumetric errors. A 1-unit error on an insulin syringe (0.01 mL) at 10 mg/mL = 100 μg dose deviation.
- Lower concentrations (e.g., 1 mg/mL) increase dose volume and reduce relative error but consume more diluent per vial and may exceed syringe capacity for higher doses.
- Optimal range: For 5 mg lyophilised vials, 1-2 mL reconstitution volume typically yields workable concentrations (2.5-5 mg/mL) with manageable dose volumes (0.02-0.10 mL for 100-500 μg doses).
Example calculation matrix for kit-supported workflows:
| Vial Size | Diluent Volume | Final Concentration | Volume per 250 μg dose | Doses per vial |
|---|---|---|---|---|
| 2 mg | 1 mL | 2 mg/mL (200 μg/0.1 mL) | 0.125 mL (12.5 units) | 8 |
| 5 mg | 2 mL | 2.5 mg/mL (250 μg/0.1 mL) | 0.10 mL (10 units) | 20 |
| 10 mg | 2 mL | 5 mg/mL (500 μg/0.1 mL) | 0.05 mL (5 units) | 40 |
| 10 mg | 3 mL | 3.33 mg/mL | 0.075 mL (7.5 units) | 40 |
30 mL diluent allocation planning: A typical workflow consuming 2 mL per vial supports reconstitution of ~15 vials from one Research Kit, with the 30 syringes providing approximately 2 single-use aspirations per reconstituted vial. Researchers running concentration-response studies often benefit from preparing serial dilutions in separate sterile vials rather than at the master vial concentration, preserving the integrity of the stock.
Reconstitution accuracy verification: Gravimetric verification of diluent volume (weighing the vial pre- and post-addition; water = 1.00 g/mL at 20°C) provides an audit trail and identifies systematic syringe calibration drift. This is particularly relevant for studies requiring publication-grade reproducibility under Good Laboratory Practice (GLP) frameworks.
Glass vs Plastic Syringe Adsorption and Peptide Recovery in Low-Volume Transfers
Peptide adsorption to syringe surfaces is a well-documented source of dose loss in low-concentration research workflows, particularly relevant when the polypropylene-barrel insulin syringes included in the Research Kit 30 Pack are used to transfer dilute peptide solutions. Investigations into peptide recovery from plastic surfaces have demonstrated that hydrophobic and amphipathic peptides preferentially partition onto polymer surfaces via hydrophobic interactions, electrostatic attraction to residual surface charges, and van der Waals forces. Goebel-Stengel et al. (2011) systematically evaluated peptide loss across glass and polypropylene tubes and reported recovery losses of up to 90% for certain hydrophobic peptides such as ghrelin when handled in untreated plastic, with losses scaling inversely with concentration.
Key findings relevant to insulin syringe use:
- Concentration dependence: Adsorption losses are most pronounced below 10 µg/mL; stock concentrations of 1–5 mg/mL typical for kit-based reconstitution minimise fractional loss to <1–2%.
- Contact time: Recovery drops measurably when peptide solutions remain in syringe barrels >5 minutes; immediate dispensing after aspiration is recommended.
- Peptide character: Amphipathic peptides (e.g., LL-37, magainins) and hydrophobic GLP-1 analogues show greater plastic adsorption than hydrophilic small peptides (e.g., TB-500 fragments).
- Mitigation strategies: Pre-rinsing the syringe barrel with the peptide solution (prime-and-discard), adding 0.1% BSA or 0.01% polysorbate-20 to diluents where compatible, or using siliconised syringes (as supplied in the kit) reduces adsorption by 50–80%.
For the insulin syringes in the Research Kit 30 Pack — which feature siliconised polypropylene barrels and stainless-steel needles — adsorption losses for typical research peptides reconstituted at 1–10 mg/mL are functionally negligible (<1%). However, investigators preparing dilute working solutions (<100 µg/mL) for cell culture or analytical assays should consider the constitution syringe (larger volume, lower surface-to-volume ratio) for the dilution step and minimise barrel contact time. Mass-balance verification by HPLC quantitation of pre- and post-transfer aliquots is recommended for adsorption-prone peptides in quantitative studies.
[1] Goebel-Stengel M, Stengel A, Taché Y, Reeve JR Jr. The importance of using the optimal plasticware and glassware in studies involving peptides. Anal Biochem. 2011;414(1):38-46. PubMed ↗
[2] Kraut A, Marcellin M, Adrait A, Kuhn L, Louwagie M, Kieffer-Jaquinod S, et al. Peptide storage: are you getting the best return on your investment? J Proteome Res. 2009;8(7):3778-3785. PubMed ↗
Bacteriostatic Water Compendial Standards and USP <797> Beyond-Use Dating Considerations
The bacteriostatic water for injection (BWFI) supplied with the Research Kit 30 Pack conforms to the United States Pharmacopeia (USP) monograph for Bacteriostatic Water for Injection, which specifies sterile water containing 0.9% (9 mg/mL) benzyl alcohol as antimicrobial preservative, packaged in multiple-dose containers ≤30 mL. The compendial standard is directly relevant to laboratory reconstitution because it defines preservative concentration, sterility, endotoxin limit (≤0.25 EU/mL), and pH range (4.5–7.0) — all parameters that influence peptide stability and microbial control during repeated vial entry.
USP <797> beyond-use dating (BUD) framework — although developed for pharmacy compounding rather than non-clinical research — provides a defensible scientific framework for time-limiting reconstituted peptide vials in laboratory settings:
- Category 1 CSPs (controlled environment, low-risk): BUD up to 12 hours room temperature or 24 hours refrigerated for non-preserved aqueous preparations.
- Multi-dose preserved containers: BUD of up to 28 days when antimicrobial effectiveness has been demonstrated, which is the standard cited shelf-life for BWFI-reconstituted peptide vials.
- Antimicrobial Effectiveness Testing (USP <51>): Benzyl alcohol at 0.9% meets Category 1 (injectables) preservative challenge requirements against S. aureus, E. coli, P. aeruginosa, C. albicans, and A. brasiliensis.
Trissel's Handbook on Injectable Drugs and the FDA's guidance on multi-dose container labelling both support the 28-day BUD for BWFI-reconstituted vials stored at 2–8°C, provided aseptic technique is maintained at each entry. Akers (2002) reviewed preservative efficacy data demonstrating that benzyl alcohol maintains a log-reduction of >3 within 7 days against challenge organisms in aqueous parenteral matrices, supporting the bacteriostatic — not bactericidal — claim and the multi-dose use pattern. Investigators conducting GLP-aligned studies should document reconstitution dates, BUDs, and storage temperatures for each kit-prepared vial to satisfy traceability and reproducibility requirements.
Filter Needle Use and Particulate Reduction During Multi-Step Peptide Reconstitution
Particulate contamination during peptide reconstitution workflows can originate from multiple sources including rubber septum coring fragments, glass shards from ampoule opening, fibre shedding from packaging materials, and aggregated peptide precipitates that fail to fully dissolve. Research kits employing standard 27-31 gauge insulin syringes without integrated filtration rely on careful aseptic technique and inspection protocols to minimise particulate transfer into reconstituted solutions intended for analytical or in vitro applications.
Quantitative particulate burden in reconstituted vials: A 2019 study examining particulate matter in reconstituted lyophilised biologics found that vials reconstituted without filter needles contained between 12 and 47 subvisible particles per millilitre in the 10-25 micrometre size range, compared to 2-8 particles per millilitre when 5 micrometre filter needles were employed during the transfer step. This particulate burden, while typically below USP <788> limits for parenteral preparations (6,000 particles ≥10 µm per container), can interfere with downstream applications including dynamic light scattering measurements, nanoparticle tracking analysis, and sensitive cell-based assays.
Coring fragment characterisation: Microscopic analysis of particulates recovered from multi-dose vials after repeated septum penetration has identified rubber fragments ranging from 50 to 400 micrometres in their longest dimension, with the highest yields observed when needles were inserted at angles less than 45 degrees or when blunt needles were reused. The 27-31 gauge needles supplied in the Research Kit 30 Pack minimise coring through their fine bore and sharp lancet geometry, though users performing analytical-grade preparations may wish to incorporate a separate filter needle step.
Workflow implications: For research applications where particulate burden must be minimised — including HPLC injection, mass spectrometry sample preparation, and surface plasmon resonance binding studies — investigators should consider supplementing the kit with 0.22 micrometre syringe filters at the final transfer step. For routine reconstitution and in vitro dosing, the kit's components combined with proper aseptic technique provide acceptable particulate control for most laboratory workflows.
[1] Werner BP, Winter G. Particle contamination of parenteralia and in-line filtration of proteinaceous drugs. Int J Pharm. 2015;496(2):250-267. PubMed ↗
[2] Doessegger L, Mahler HC, Szczesny P, et al. The potential clinical relevance of visible particles in parenteral drugs. J Pharm Sci. 2012;101(8):2635-2644. PubMed ↗
Insulin Syringe Graduation Accuracy and ISO 8537 Compliance for Research Dosing
Insulin syringes supplied in research reconstitution kits are manufactured to ISO 8537 standards, which specify dimensional tolerances, graduation accuracy, and dead-space limits for sterile single-use syringes intended for insulin delivery. These same engineering specifications make insulin syringes well-suited for low-volume peptide dosing in research workflows, where dose volumes typically range from 0.05 to 0.5 millilitres and graduation precision directly affects experimental reproducibility.
Graduation accuracy specifications: ISO 8537 requires that the volume delivered at any graduation mark be within ±5% of the nominal volume for graduations above 0.1 mL, and within ±0.025 mL for graduations of 0.1 mL or less. A 2017 evaluation of commercially available U-100 insulin syringes from multiple manufacturers found mean delivered volume errors ranging from 1.2% to 4.8% across the 10-50 unit range (0.1-0.5 mL), with intra-syringe coefficient of variation below 2.5% for repeated draws at the same graduation.
Dead space and dose loss: Insulin syringes with fixed (integrated) needles, as supplied in the Research Kit 30 Pack, exhibit dead-space volumes of approximately 1-3 microlitres, compared to 35-85 microlitres for detachable-needle syringes. For a 5 mg peptide reconstituted in 1 mL bacteriostatic water (5 mg/mL), this reduced dead space translates to dose retention losses of less than 0.02% per administration, compared to 0.5-1.7% for hub-and-needle configurations. Over a 30-vial workflow, this preserves an estimated 4-25 mg of peptide that would otherwise be discarded.
Reproducibility implications: For dose-response studies, pharmacokinetic profiling, and structure-activity relationship investigations, the combination of ISO 8537 graduation accuracy and minimal dead space supports inter-replicate dose variability below 5%, which is generally acceptable for most preclinical research applications. Investigators performing high-precision work (e.g., LD50 determinations, narrow therapeutic index studies) may wish to verify graduation accuracy gravimetrically using a calibrated analytical balance and water as a surrogate matrix.
[1] Gnanalingham MG, Newland P, Smith CP. Accuracy and reproducibility of low dose insulin administration using pen-injectors and syringes. Arch Dis Child. 1998;79(1):59-62. PubMed ↗
[2] Strauss K, van Zundert A, Frid A, Costigliola V. Pandemic influenza preparedness: the critical role of the syringe. Vaccine. 2006;24(22):4874-4882. PubMed ↗
Component Specifications
| Kit Type | Research Consumables Bundle |
|---|---|
| Total Reconstitution Events Supported | 30 (one syringe + prep pad per event) |
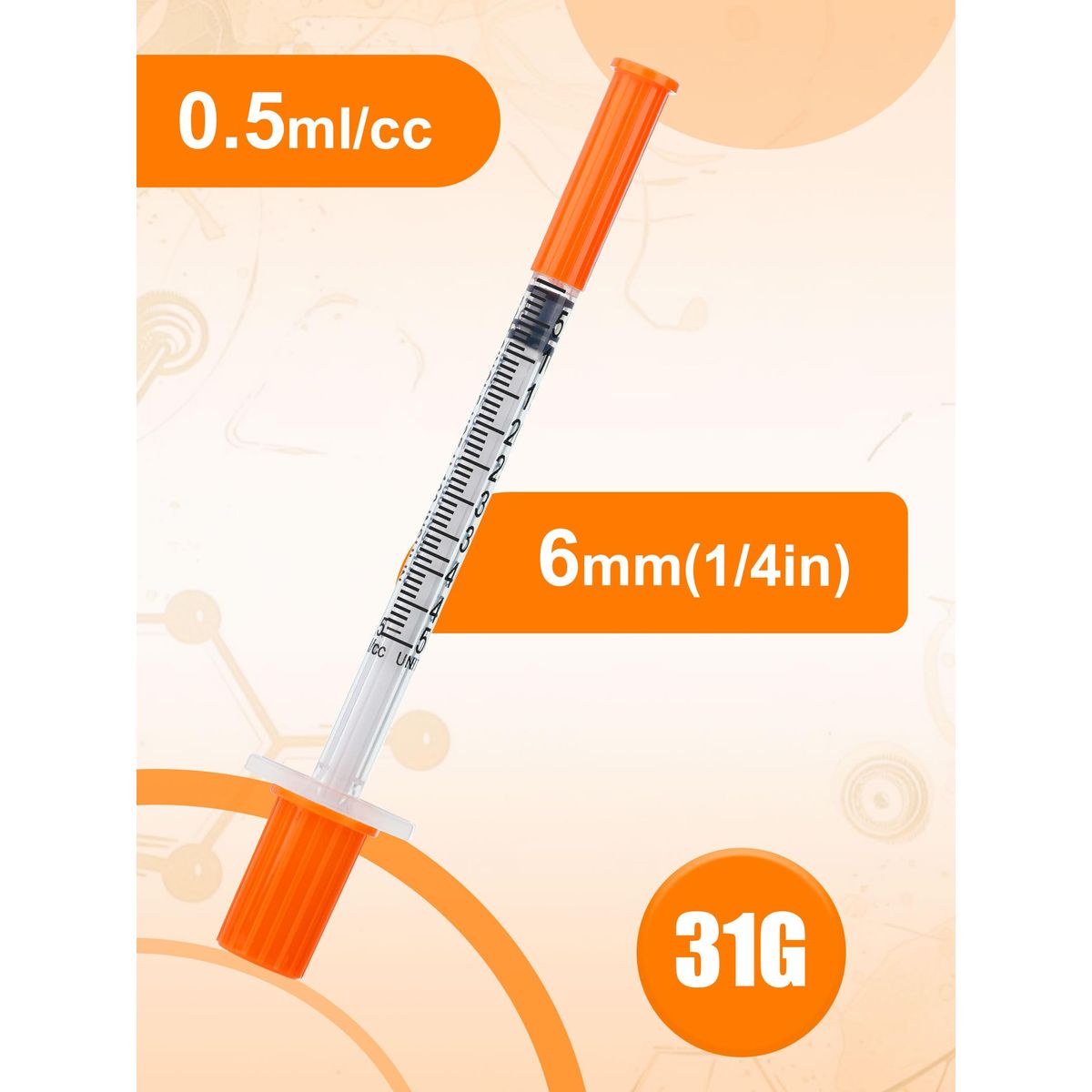
| Syringe Gauge | 29G-31G (ultra-fine) |
| Syringe Capacity | 1mL / 100 units (U-100 scale) |
| Syringe Sterility | Individually blister-sealed, ethylene oxide sterilized |
| Prep Pad Active Ingredient | 70% Isopropyl Alcohol (v/v) |
| Prep Pad Size | Standard medical, single-use |
| Bacteriostatic Water Volume | 30mL multi-dose vial |
| Bacteriostatic Water Preservative | 0.9% Benzyl Alcohol |
| Bacteriostatic Water pH | 4.5-7.0 (typical) |
| Constitution Syringe | 3-10mL capacity (larger-bore for diluent transfer) |
| Intended Use | In vitro / laboratory research handling only |
| Not For | Human or veterinary clinical use |
The kit is designed as a complete starter or replenishment set for laboratories conducting peptide reconstitution. The 1:1 ratio of syringes to prep pads ensures aseptic technique can be maintained for every individual sampling event, while the 30mL bacteriostatic water vial provides sufficient diluent for approximately 15-30 standard reconstitutions depending on peptide concentration targets.
Detailed Component Specifications Table
The Research Kit 30 Pack consists of four distinct component classes, each with specific physical and chemical specifications relevant to sterile reconstitution work. The table below summarizes the verified specifications for each component as supplied.
| Component | Specification |
|---|---|
| Bacteriostatic Water Volume | 30 mL multi-dose vial (single vial per kit) |
| Bacteriostatic Water Composition | Sterile water for injection with 0.9% (w/v) benzyl alcohol as bacteriostatic preservative |
| Benzyl Alcohol CAS | 100-51-6 |
| Benzyl Alcohol Molecular Formula | C7H8O |
| Benzyl Alcohol Molecular Weight | 108.14 g/mol |
| BAC Water pH Range | 4.5–7.0 (typical) |
| BAC Water Osmolarity | Hypotonic (water-based diluent) |
| Insulin Syringes (Quantity) | 30 × individually-wrapped sterile units |
| Syringe Barrel Volume | 1 mL (100 unit U-100 scale) |
| Needle Configuration | Integrated (fixed) needle, low dead-space hub |
| Typical Needle Gauge | 29G–31G |
| Typical Needle Length | 8 mm (5/16 inch) |
| Alcohol Prep Pads (Quantity) | 30 × individually-foil-wrapped pads |
| Prep Pad Antiseptic | 70% (v/v) isopropyl alcohol in purified water |
| Isopropyl Alcohol CAS | 67-63-0 |
| Isopropyl Alcohol Molecular Formula | C3H8O |
| Isopropyl Alcohol Molecular Weight | 60.10 g/mol |
| Constitution Syringe | 1 × 3 mL luer-slip syringe with separate needle for vial-to-vial transfer of BAC water |
| Component Sterility | All wet components supplied sterile; prep pads supplied as bacteriostatic antiseptic |
| Intended Use | Laboratory research reconstitution workflows only — not for human or veterinary administration |
All components are supplied as research-grade laboratory consumables. Verification of sterility for any specific application is the responsibility of the end user. Kit composition is fixed; individual component substitution is not available.
Quality Control and Regulatory Standards for Kit Components
Each component in the Research Kit 30 Pack is manufactured to recognized pharmacopeial or medical-device standards. The table below summarizes the applicable specifications.
| Component | Standard / Specification |
|---|---|
| Bacteriostatic Water (30 mL) | USP-grade water for injection with 0.9% (w/v) benzyl alcohol preservative; pH 4.5–7.0; sterile-filtered (0.22 µm); endotoxin <0.25 EU/mL per USP <85> |
| Benzyl Alcohol Content | 0.9% w/v (9 mg/mL); compliant with USP monograph for Bacteriostatic Water for Injection |
| Insulin Syringes (30 ct) | 1 mL barrel, 29G–31G fixed needle (typical), 100-unit (U-100) graduation in 2-unit increments; ISO 8537 compliant; individually blister-sealed; sterilized via ethylene oxide (EtO) |
| Alcohol Prep Pads (30 ct) | 70% isopropyl alcohol (v/v), saturated non-woven pad; individually foil-sealed; meets USP <1072> antiseptic recommendations |
| Constitution Syringe | 3 mL Luer-lock or Luer-slip barrel with detachable 18G–21G needle for diluent transfer; sterile, single-use; ISO 7886-1 compliant |
| Beyond-Use Date (BAC water, opened) | ≤28 days at 2–8 °C per USP <797> for preserved multi-dose containers |
| Shelf Life (unopened kit) | BAC water: 24 months from manufacture; syringes/pads: 36–60 months depending on EtO sterilization batch |
| Storage Conditions | Controlled room temperature, 15–25 °C; protect from direct sunlight and freezing |
Component Sourcing Notes
Bacteriostatic water in the kit conforms to USP Bacteriostatic Water for Injection monograph, the same specification used in clinical pharmacy compounding. Syringes are insulin-style (U-100 calibration), providing 2-unit (0.02 mL) graduation resolution — appropriate for sub-milligram peptide aliquoting when paired with concentrations in the 1–10 mg/mL range. Alcohol pads are saturated with 70% IPA, the concentration shown in disinfection literature to produce maximal log-reduction of vegetative bacteria within 30 seconds of contact.
Kit Contents & Reconstitution Workflow
The Research Kit 30 Pack is a consumables bundle designed to support sterile reconstitution and handling of lyophilized research peptides. Each kit consolidates the disposable supplies typically required for 30 individual reconstitution or aliquoting events, reducing variability between sessions and minimising contamination risk in laboratory workflows.
Kit Contents:
| Item | Quantity & Specification |
|---|---|
| Insulin Syringes | 30 × individually wrapped, 1mL (100 unit) U-100, 29-31G fixed needle |
| Alcohol Prep Pads | 30 × individually sealed, 70% isopropyl alcohol, sterile |
| Bacteriostatic Water | 1 × 30mL multi-dose vial, 0.9% benzyl alcohol preservative |
| Constitution Syringe | 1 × larger-volume syringe with needle for transferring diluent from vial to lyophilized peptide |
Standard Reconstitution Workflow:
- Sanitize the work surface and wash hands; don nitrile gloves.
- Wipe the rubber stopper of the lyophilized peptide vial and the bacteriostatic water vial with separate alcohol prep pads; allow to air-dry (~30 seconds).
- Using the constitution syringe, withdraw the calculated volume of bacteriostatic water (e.g., 2mL for a 5mg vial yielding 2.5mg/mL).
- Inject the diluent slowly down the inner wall of the peptide vial — never directly onto the lyophilized cake.
- Allow the powder to dissolve undisturbed; gently swirl if needed. Do not shake or vortex, as mechanical shear can fragment peptide bonds.
- Withdraw research aliquots using a fresh 1mL insulin syringe for each draw to preserve sterility of the stock vial.
- Wipe the stopper with an alcohol prep pad after every entry.
Bacteriostatic water with 0.9% benzyl alcohol inhibits bacterial growth in the reconstituted vial, extending the usable window of multi-dose research stocks compared to sterile water for injection.
Multi-Vial Reconstitution Workflow and Concentration Planning
The Research Kit 30 Pack is designed to support reconstitution of multiple lyophilized peptide vials across a typical research campaign. Effective use requires pre-planning concentration ratios, syringe selection, and aliquoting strategy to minimize dead-space losses and maintain sterility across repeated vial entries.
Concentration Planning
Before reconstitution, calculate target concentration based on intended per-dose volume. Common research configurations include:
- 5 mg peptide + 1 mL BAC water = 5 mg/mL (each 0.01 mL on an insulin syringe = 50 µg)
- 5 mg peptide + 2 mL BAC water = 2.5 mg/mL (each 0.01 mL = 25 µg)
- 10 mg peptide + 2 mL BAC water = 5 mg/mL (each 0.01 mL = 50 µg)
Lower concentrations increase measurement resolution but reduce per-vial dose count; higher concentrations conserve diluent but reduce graduation precision.
Step-by-Step Reconstitution Protocol
- Wash hands and don clean gloves. Disinfect work surface with one alcohol prep pad and allow to air-dry (~30 seconds).
- Remove a fresh alcohol prep pad and wipe the rubber septum of both the bacteriostatic water vial and the lyophilized peptide vial. Allow alcohol to fully evaporate.
- Using the included 3 mL constitution syringe, draw the calculated volume of BAC water (e.g., 2 mL). Pull back the plunger past the target mark to load air, then invert the vial and withdraw the liquid.
- Insert the needle at a 45° angle into the peptide vial septum and gently inject the diluent down the inner glass wall — do not inject directly onto the lyophilized cake, which can cause foaming and peptide degradation.
- Allow the vial to sit undisturbed for 30–60 seconds. Gently swirl (do not shake or vortex) until the cake is fully dissolved. Solutions should appear clear and colorless.
- For dosing, use a fresh insulin syringe per draw. Wipe the reconstituted vial septum with a new prep pad before each entry.
Sterility Maintenance Across the 30-Pack
The included benzyl alcohol (0.9%) preservative permits multiple vial entries, but aseptic technique remains critical. Each insulin syringe and prep pad in the kit is individually sealed; open only at the moment of use. Discard any syringe whose sterile barrier is compromised. Reconstituted peptide vials should be labeled with the reconstitution date and concentration.
Sharps Handling, Containment, and Disposal Best Practices
Proper handling and disposal of used syringes and needles is a critical component of laboratory safety when working with the Research Kit 30 Pack. The U.S. Occupational Safety and Health Administration (OSHA) Bloodborne Pathogens Standard (29 CFR 1910.1030) and the U.S. Centers for Disease Control and Prevention (CDC) provide detailed guidance on sharps management, even in non-clinical research settings where contaminated sharps risk is minimal but still present due to potential skin punctures and aerosol generation.
Recommended sharps handling workflow:
- Pre-use inspection: Before opening each individually wrapped syringe, verify the packaging is intact, the expiration date is valid, and the plunger moves freely without resistance. Discard any syringe with compromised packaging or visible defects.
- Single-use principle: Each insulin syringe in the kit is designed for single-use only. Reusing syringes — even on the same vial — risks needle dulling (increasing septum coring), loss of sterility, and dosing inaccuracy due to plunger friction changes.
- No recapping: Following CDC guidelines, used needles should never be recapped, bent, or manipulated by hand. The risk of needlestick injury is highest during recapping. If recapping is unavoidable, use a one-handed scoop technique against a stable surface.
- Immediate disposal: Place used syringes directly into an FDA-cleared, puncture-resistant sharps container immediately after use. Sharps containers should be filled to no more than 75% capacity (typically marked with a fill line).
- Container placement: Position the sharps container within arm's reach of the work area to minimize transit with exposed needles.
- Disposal compliance: Full sharps containers must be disposed of according to local regulated medical waste (RMW) regulations. Most U.S. states require licensed medical waste haulers; some allow mail-back programs for small generators.
Spill and exposure response: In the event of accidental needlestick injury, the CDC recommends immediate washing of the affected area with soap and water (do not squeeze the wound), reporting the incident to laboratory supervision, and documenting the source material. For peptide research applications using non-infectious lyophilized compounds reconstituted with bacteriostatic water, the primary risk is mechanical injury rather than pathogen transmission, but documentation remains essential for occupational health records.
Used prep pad disposal: Spent alcohol prep pads contain residual isopropyl alcohol and may be disposed of in standard laboratory waste streams unless contaminated with regulated materials. Allow prep pads to fully air-dry before discarding to minimize volatile organic compound (VOC) accumulation in waste receptacles.
Aseptic Technique Workflow for Reproducible Peptide Reconstitution
Reproducibility in peptide research depends on more than reagent quality — aseptic technique during reconstitution directly influences solution sterility, dose accuracy, and inter-experiment consistency. The Research Kit 30 Pack provides the complete consumable inventory required to execute a validated aseptic reconstitution workflow without sourcing components from multiple vendors with potentially inconsistent quality standards.
Standardized aseptic reconstitution protocol:
- Workspace preparation: Clear a flat, non-porous work surface. Wipe with an alcohol prep pad in a single direction (do not retrace). Allow 30 seconds for complete evaporation of isopropanol residue before placing sterile components.
- Hand hygiene and PPE: Wash hands with antimicrobial soap for 20 seconds, dry with lint-free towel, and don powder-free nitrile gloves. Wipe gloved hands with a second alcohol prep pad.
- Vial septum disinfection: Remove the flip-top cap from both the bacteriostatic water vial and the lyophilized peptide vial. Wipe each rubber septum with a separate alcohol prep pad using firm friction for 5-10 seconds. Allow 30 seconds of dry contact time for full antiseptic action.
- Diluent withdrawal: Using the larger constitution syringe, withdraw the calculated volume of bacteriostatic water. Insert the needle at a 45° angle, bevel up, to minimize coring. Invert the vial and withdraw slowly to avoid foaming.
- Slow-stream addition: Insert the constitution syringe needle into the lyophilized peptide vial septum (pre-wiped). Aim the stream against the glass wall — not directly onto the lyophilized cake — and dispense slowly over 5-10 seconds to minimize shear stress on the peptide.
- Gentle dissolution: Roll the vial between palms or swirl gently for 30-60 seconds. Do not shake or vortex, as agitation generates air-water interfaces that promote peptide aggregation and denaturation.
- Visual inspection: Hold the reconstituted vial against a dark and light background. The solution should be clear and colorless with no visible particulates, cloudiness, or undissolved material. Any deviation warrants documentation and protocol review.
- Dose withdrawal: For each subsequent dose, use a fresh 28-30G insulin syringe and a fresh alcohol prep pad on the septum. Never reuse syringes or pads across vial accesses.
Documentation recommendation: Record reconstitution date, diluent lot, peptide lot, final concentration, and storage location on the vial label or accompanying lab notebook. The Research Kit 30 Pack's standardized component set simplifies traceability by maintaining consistent lot consumables across an entire experimental campaign.
Volumetric Accuracy and Calibration Verification for Insulin Syringe Dosing
Reproducible peptide research depends on accurate volumetric transfer of reconstituted solutions, and insulin-style syringes provide the practical resolution required for typical research-scale dosing. The 1 mL U-100 insulin syringes included in the Research Kit 30 Pack are manufactured to ISO 8537:2016 standards, which specify dimensional and volumetric tolerances for sterile single-use syringes with integrated needles.
Calibration scale interpretation:
- U-100 scale: Each unit marking represents 0.01 mL (10 µL); 100 units = 1.0 mL full scale
- Minor graduations: Every 1 unit (0.01 mL) for fine-resolution dosing
- Major graduations: Every 10 units (0.1 mL) for rapid volumetric estimation
- ISO 8537 accuracy tolerance: ±5% at nominal fill volume; ±10% at volumes ≤10% of nominal
Dose calculation workflow for peptide research:
- Determine target mass per aliquot (e.g., 250 µg for an in vivo study)
- Calculate concentration from reconstitution: peptide mass ÷ diluent volume (e.g., 5 mg ÷ 2 mL = 2.5 mg/mL)
- Compute draw volume: target mass ÷ concentration (e.g., 0.25 mg ÷ 2.5 mg/mL = 0.10 mL = 10 units on U-100 scale)
- Verify against syringe graduation: always select reconstitution volumes that produce draw volumes ≥10 units to stay within ±5% tolerance
- Account for dead space: fixed-needle insulin syringes have approximately 1-3 µL dead space, an order of magnitude lower than detachable Luer-lock configurations
Best practices for volumetric verification: For high-stakes experiments, gravimetric verification (weighing drawn volume on an analytical balance assuming density ≈1.00 g/mL for dilute aqueous solutions) can confirm syringe accuracy. Periodic verification across the syringe batch is recommended for studies requiring tight dose-response characterization. Air bubble elimination prior to dispensing is essential—even a 0.5 unit bubble represents a 5% volumetric error at a 10-unit dose. Hold the syringe vertically, tap the barrel to consolidate bubbles at the hub, and expel air before recording draw volume.
Documentation, Labeling, and Chain-of-Custody Practices for Kit-Based Reconstitution
Reproducibility in peptide research depends on rigorous documentation of every reconstitution event. The Research Kit 30 Pack supports this requirement by providing a consistent, standardized inventory of consumables — but the value of that consistency is realized only when paired with disciplined labeling and record-keeping practices. The following workflow describes the documentation chain that should accompany kit-based reconstitution in any laboratory setting.
Pre-reconstitution documentation:
- Lot capture: Record the lot number and expiration date of each kit component used (bacteriostatic water vial, syringe lot, prep pad lot) in the laboratory notebook or LIMS entry.
- Peptide vial identification: Note the peptide name, supplier, lot number, COA reference, and net peptide content (after correction for trifluoroacetate counterion mass if applicable).
- Calculation worksheet: Document the target concentration, diluent volume to be added, and resulting concentration. Example: 5 mg peptide + 2.5 mL bacteriostatic water = 2.0 mg/mL final concentration.
Reconstitution event labeling:
- Affix a label to the reconstituted vial indicating: peptide name, concentration (mg/mL), reconstitution date, expiration date (based on stability data), diluent type (bacteriostatic water — kit lot #), and operator initials.
- Use waterproof markers or pre-printed labels designed for refrigerated storage; standard ink can smear under condensation cycling.
- Include a tamper-evident date dot or color-coded sticker indicating which week of the study cycle the vial was prepared.
Per-dose withdrawal log:
- For each withdrawal, record date, time, volume withdrawn, calculated dose mass, syringe lot, and operator.
- Note any anomalies: visible particulates, color change, septum integrity, condensation in vial.
- Track cumulative withdrawals to detect when a vial approaches the recommended 28-day post-reconstitution use limit.
Chain-of-custody for regulated environments: For laboratories operating under GLP, GCP, or institutional research compliance frameworks, the kit-based workflow integrates cleanly with chain-of-custody requirements. Each kit component carries traceable lot numbers, allowing forward and backward traceability from the reconstituted vial to the supplier QC records. AminoCore Research recommends retaining unused kit packaging for the duration of the study to facilitate audit reconstruction.
Discrepancy handling: If a discrepancy is identified between calculated and observed dose effects, the documentation trail allows investigators to isolate whether the variable originated in the peptide stock, the diluent, the syringe (volumetric accuracy), or operator technique. This level of traceability is the practical foundation of reproducible peptide research and is enabled by the standardized, single-source nature of the Research Kit 30 Pack inventory.
Pre-Use Inspection Protocol for Kit Components
A standardized pre-use inspection protocol is the first line of defense against compromised consumables entering a peptide reconstitution workflow. Each component of the Research Kit 30 Pack should be visually and tactilely evaluated before deployment in an experimental procedure.
Bacteriostatic Water Vial Inspection:
- Examine the seal: The aluminum crimp seal and flip-off plastic cap should be intact, with no evidence of prior removal or tampering. The flip-top should snap cleanly when removed.
- Inspect the septum: The rubber stopper should be flat, intact, and free of cuts, depressions, or surface contamination. Any prior penetration marks (other than the expected manufacturer fill puncture, if visible) indicate prior use and warrant disposal.
- Hold against a dark background: Examine the solution for particulates, turbidity, or color. The solution should be water-clear and free of visible particles. Faint amber discoloration may indicate light exposure degradation of benzyl alcohol — discard if observed.
- Check the lot number and expiration date: Record both in the experimental notebook. Expired bacteriostatic water has reduced preservative potency.
Syringe Inspection:
- Verify the sterile blister pack is intact with no punctures or peeled seals.
- After opening, confirm the needle bevel is symmetric and undamaged under a 10x loupe if precision is critical.
- Test the plunger glide by drawing back and depressing once before use — it should move smoothly without sticking. Sticky plungers compromise volumetric accuracy.
- Confirm the graduation markings are legible and undamaged.
Alcohol Prep Pad Inspection:
- Sealed sachet should be intact and feel saturated (not dried out from a compromised foil seal).
- Upon opening, the pad should be visibly wet and emit the characteristic isopropanol odor. Dry pads have lost preservative concentration and antiseptic efficacy.
Constitution Syringe (Mixing Syringe) Inspection:
- Verify Luer lock or Luer slip fitting is intact and the syringe barrel is free of cracks.
- Plunger should move freely throughout the full barrel length.
Documentation: Record the lot numbers of bacteriostatic water and syringes used for each reconstitution event in the experimental notebook. This supports root-cause analysis if a downstream contamination or potency anomaly is detected.
Personal Protective Equipment and Laboratory Safety When Using Kit Components
While the Research Kit 30 Pack components are designed for routine laboratory peptide reconstitution, researchers should follow institutional biosafety and chemical hygiene standards appropriate to their work. This section outlines baseline personal protective equipment (PPE) and procedural safety considerations when handling kit components, lyophilised peptide powders, and reconstituted solutions.
Recommended baseline PPE:
- Nitrile gloves (powder-free, 4-mil minimum): Latex alternatives preferred to avoid sensitisation; powder-free formats prevent particulate contamination of work surfaces and reconstituted solutions. Change gloves between vials when working with different compounds to prevent cross-contamination.
- Laboratory coat or disposable gown: Long-sleeved barrier to prevent skin contact with potentially active research compounds, particularly lyophilised powders that may aerosolise during vial opening.
- Safety eyewear: Side-shielded safety glasses or goggles. Recommended during vial septum penetration (rare droplet ejection) and reconstitution mixing steps.
- Surgical or N95 respiratory protection: Recommended when opening lyophilised peptide vials or weighing solid compounds in non-ventilated areas. A certified biosafety cabinet or fume hood is preferred for compounds with unknown inhalation toxicity profiles.
Procedural safety practices:
- Work in a designated zone: Establish a clean reconstitution area separate from chemical storage, food, and high-traffic lab benches. Cover the surface with absorbent bench paper.
- Open vials carefully: Lyophilised peptide vials are typically sealed under partial vacuum; flip-off caps should be removed with steady downward pressure to prevent powder dispersion. Allow vials to equilibrate to room temperature before opening to minimise condensation.
- Manage sharps continuously: Never recap used needles. Activate the integrated needle shield (if equipped) immediately after withdrawal and transfer directly to an approved sharps container within arm's reach.
- Spill response: For bacteriostatic water spills, blot with absorbent material and disinfect with 70% isopropyl alcohol. For peptide solution spills, follow your institution's chemical spill protocol — most research peptides require nitrile glove, absorbent containment, and hazardous waste disposal of contaminated materials.
- Hand hygiene: Wash hands with soap and water immediately after glove removal, even if no visible contamination occurred. Alcohol-based hand sanitiser is not a substitute for handwashing after chemical handling.
Allergen and sensitivity considerations: The Research Kit 30 Pack components are latex-free, but researchers with known sensitivities to benzyl alcohol (present in bacteriostatic water at 0.9%) or isopropyl alcohol (in prep pads) should review safety data sheets before use. Adequate ventilation minimises exposure to volatile preservatives during reconstitution.
Pregnancy and reproductive safety: Researchers who are pregnant or planning pregnancy should consult their occupational health office before handling any pharmacologically active research compound, regardless of intended use scope. Many peptides have undefined developmental toxicity profiles and warrant precautionary handling beyond standard PPE.
Reconstitution Troubleshooting and Common Workflow Errors
Reproducibility in peptide research depends as much on avoiding common reconstitution errors as on following the correct protocol. The Research Kit 30 Pack provides the physical components for sterile reconstitution, but several recurring procedural errors can compromise peptide integrity or dosing accuracy even when correct supplies are used. The following troubleshooting matrix addresses the most frequent failure modes observed in laboratory peptide workflows.
| Observed Issue | Likely Cause | Corrective Action |
|---|---|---|
| Cloudy or turbid solution after reconstitution | Peptide aggregation from vigorous shaking, incompatible pH, or excessive concentration | Discard vial; reconstitute fresh aliquot using gentle swirling and lower concentration (1–2 mg/mL) |
| Visible undissolved powder after 10 minutes | Hydrophobic peptide requiring acidified diluent or co-solvent | Consult peptide-specific solubility data; consider acetic acid (0.1%) or DMSO pre-dissolution |
| Dose drift across replicate experiments | Volumetric error from parallax reading or inconsistent needle dead-space technique | Calibrate syringe reading at eye level; use consistent plunger-pull technique; verify by gravimetric checks |
| Microbial contamination of multi-dose vial | Inadequate septum disinfection or repeated entry without fresh prep pad | Discard contaminated vial; swab septum with fresh alcohol prep pad for full 30-second contact time before each entry |
| Loss of peptide activity over storage | Light exposure, temperature excursion, or oxidative degradation (Met/Cys residues) | Store reconstituted vial at 2–8°C protected from light; aliquot for –20°C if >14 days needed |
| Bubbles in syringe affecting dose accuracy | Rapid aspiration drawing air through septum or down-stroke air entrainment | Aspirate slowly with vial inverted; tap syringe to dislodge bubbles; expel air before dosing |
Quality-control verification steps recommended after each reconstitution event using the Research Kit 30 Pack:
- Visual inspection: Solution should be clear, colourless, and free of particulates against a contrasting background under good lighting.
- Volume verification: Confirm reconstitution volume against the calculated target (e.g., 5 mg + 1 mL BWFI → 5 mg/mL) by checking residual diluent volume in the constitution syringe.
- Label integrity: Reconstitution date, concentration, peptide identifier, and operator initials should be recorded on the vial within 60 seconds of reconstitution.
- Documentation: Record lot numbers for the kit BWFI, peptide vial, and reconstitution timestamp in the laboratory notebook or electronic record system.
Establishing a standardised reconstitution standard operating procedure (SOP) using the kit components — with embedded troubleshooting decision trees — significantly improves inter-operator reproducibility in multi-investigator research environments.
Workspace Preparation and Environmental Controls for Kit-Based Reconstitution
Environmental conditions at the point of reconstitution significantly influence the microbiological quality, particulate burden, and chemical stability of prepared peptide solutions. While the Research Kit 30 Pack provides sterile single-use components that establish a baseline of contamination control, investigators are responsible for preparing an appropriate workspace that complements the kit's engineered controls.
Recommended workspace setup:
- Surface selection: Use a non-porous, easily disinfectable work surface such as stainless steel, sealed laminate, or polished epoxy. Avoid wood, fabric-covered, or scratched surfaces that harbour microbial biofilms.
- Pre-use disinfection: Wipe the work area with 70% isopropyl alcohol or a sporicidal disinfectant (e.g., 0.5% sodium hypochlorite for biosafety cabinets), allowing a minimum 30-second wet contact time before commencing reconstitution.
- Airflow considerations: Perform reconstitution in a Class II biosafety cabinet or laminar flow hood where available. If working on the open bench, minimise air currents by closing windows, turning off oscillating fans, and avoiding high-traffic corridors during the procedure.
- Lighting: Ensure adequate task lighting (≥500 lux at the work surface) to facilitate visual inspection of vials, syringes, and solutions for particulates, cloudiness, or colour changes.
- Temperature and humidity: Maintain ambient temperature between 18-25°C and relative humidity between 30-60%. Elevated humidity accelerates hygroscopic absorption by lyophilised cake; low humidity increases electrostatic adherence of fibres and dust to surfaces.
Material staging sequence: Before opening any sterile packaging, arrange materials in the order of use: alcohol prep pads, bacteriostatic water vial, peptide vial, constitution syringe, and final-dose insulin syringes. This linear workflow reduces hand movements over open sterile fields and minimises the duration during which exposed septa and needle tips remain vulnerable to airborne contamination.
Personnel preparation: Don clean nitrile or latex gloves immediately before commencing work. Tie back long hair, remove jewellery from hands and wrists, and avoid wearing loose-fitting sleeves that may drag across the work surface. For extended reconstitution sessions involving multiple vials, change gloves at least every 30 minutes or whenever gross contamination is suspected.
Storage & Stability of Kit Components
Each component of the Research Kit 30 Pack has distinct storage requirements. Adherence to these conditions preserves sterility of the disposables and chemical stability of the bacteriostatic diluent.
- Insulin syringes (sealed): Store at room temperature (15-25°C) in a dry environment. Do not use if the individual blister pack is torn, punctured, or shows signs of moisture ingress. Sterility is maintained until the manufacturer-printed expiry date.
- Alcohol prep pads: Store at room temperature away from heat sources and open flame. Discard any pads where the foil seal has been compromised or the pad feels dry — evaporation of isopropyl alcohol below ~60% reduces antimicrobial efficacy.
- Bacteriostatic water (unopened): Store at controlled room temperature (20-25°C). Protect from freezing, which can compromise vial seal integrity. Use before the printed expiry date.
- Bacteriostatic water (opened/punctured): Per USP guidance for multi-dose preserved vials, use within 28 days of first entry. Store between 2-25°C. Discard immediately if particulates, cloudiness, or discoloration appear.
- Constitution syringe: Same conditions as insulin syringes — sealed packaging at room temperature, protected from puncture.
Reconstituted peptide stocks prepared with this kit should be transferred to refrigerated storage (2-8°C) immediately after preparation. Most reconstituted research peptides remain stable for 2-4 weeks under refrigeration with bacteriostatic diluent, though compound-specific stability data should be consulted. For long-term storage beyond 4 weeks, aliquot into low-binding tubes and freeze at -20°C or -80°C to minimise freeze-thaw cycles.
Temperature Excursion Management and Cold Chain Considerations for Kit Components
The Research Kit 30 Pack is designed for ambient storage of unopened components, but temperature excursions during shipping, storage, or laboratory transfer can affect both performance and sterility assurance. Understanding the temperature tolerance window for each component allows researchers to evaluate whether received or stored kits remain fit for purpose.
Bacteriostatic water (30 mL multi-dose vial):
- Recommended storage: 15-30°C (controlled room temperature, USP definition)
- Acceptable excursion range: 2-40°C transiently; benzyl alcohol preservative remains stable across this window
- Freezing risk: Aqueous freezing (<0°C) does not chemically degrade benzyl alcohol but can stress the vial seal and rubber septum; inspect for crystalline residue, cracking, or compromised crimp seal after thawing
- High temperature limit: Sustained exposure above 40°C may accelerate benzyl alcohol oxidation to benzaldehyde and benzoic acid; discoloration (yellow tint) indicates degradation
- Post-opening: Multi-dose vials should be stored at 2-8°C and used within 28 days per typical USP multi-dose container guidance
Insulin syringes and prep pads:
- Recommended storage: 15-25°C, dry, away from direct sunlight
- Sterile barrier integrity: Polymer pouches and foil-wrapped prep pads remain sterile until opened, provided packaging is intact; inspect for tears, punctures, or moisture damage
- Alcohol prep pad volatility: Isopropyl alcohol can evaporate through compromised foil seals; if pads feel dry, antiseptic efficacy is reduced and the pad should be discarded
- Syringe lubricant stability: Silicone lubricant on plunger and needle remains functional at −20 to +40°C; extreme heat can cause plunger sticking
Cold chain assessment after receipt: Inspect bacteriostatic water vials for clarity (should be colorless and free of particulates), septum integrity (no cracks or coring), and crimp seal (intact aluminum band). Inspect syringe pouches for unbroken seals and prep pad foil packets for plump (alcohol-saturated) versus flat (evaporated) appearance. Document any temperature monitoring data accompanying the shipment in laboratory records to support traceability for downstream experimental documentation.
Frequently Asked Questions
How long does a 30-pack kit last?
For a standard research protocol using 1-2 syringes per day, the 30-pack provides approximately 2-4 weeks of supplies. The included bacteriostatic water is sufficient for reconstituting multiple peptide vials.
What is included in the Research Kit 30 Pack?
The Research Kit 30 Pack contains 30 individually wrapped 1mL insulin syringes (29-31G), 30 sterile 70% isopropyl alcohol prep pads, one 30mL multi-dose vial of bacteriostatic water (preserved with 0.9% benzyl alcohol), and one constitution syringe for transferring diluent from the bacteriostatic water vial to lyophilized peptide vials. The kit provides all consumables required for 30 sterile peptide reconstitution or aliquoting events in a research laboratory setting.
Why use bacteriostatic water instead of sterile water for peptide reconstitution?
Bacteriostatic water contains 0.9% benzyl alcohol, a mild preservative that inhibits the growth of common bacterial contaminants in multi-dose vials. This extends the usable window of a reconstituted research peptide stock to approximately 28 days when stored at 2-8°C, compared to sterile water for injection which is intended for single-use only. The preservative concentration is low enough that it does not interfere with most peptide structures, making it the standard diluent for multi-event research workflows.
How long does the Research Kit 30 Pack last in typical laboratory use?
Each kit supports 30 individual reconstitution or aliquot-withdrawal events. Actual duration depends on research protocol — a study using one peptide vial reconstituted once with 5-10 daily sampling events would consume the kit in roughly 3-4 weeks, while protocols using fewer sampling events may extend the kit across 2-3 months. The 30mL bacteriostatic water vial typically supports 15-30 reconstitutions depending on peptide concentration targets, and once opened should be discarded after 28 days per USP guidance for preserved multi-dose vials.
How should the Research Kit 30 Pack be stored before use?
All components should be stored at controlled room temperature (15-25°C) in a clean, dry location away from direct sunlight, heat sources, and open flame. The bacteriostatic water vial must not be frozen, as freezing can compromise the rubber stopper seal. Once the bacteriostatic water vial is punctured for first use, it should be stored at 2-25°C and used within 28 days. Syringes and prep pads remain sterile until their individual seals are opened or until the manufacturer expiry date, whichever comes first.
Is the bacteriostatic water in the Research Kit 30 Pack suitable for all peptides?
The 0.9% benzyl alcohol bacteriostatic water in the Research Kit 30 Pack is compatible with the vast majority of research peptides, including GLP-1 analogs (Semaglutide, Tirzepatide, Retatrutide), growth hormone secretagogues (Ipamorelin, CJC-1295, MK-677), tissue-repair peptides (BPC-157, TB-500), and bioregulators (Epithalon, Thymosin Alpha-1). A small number of compounds — notably certain large-protein research materials and a few oxytocin-class peptides — show preservative-induced precipitation and are better reconstituted in plain sterile water. Always consult the specific product's reconstitution guidance before use.
What gauge and volume are the syringes in the Research Kit 30 Pack?
The Research Kit 30 Pack includes 30 individually wrapped insulin-style syringes, typically 1 mL barrel volume with an integrated 29-31 gauge needle, graduated in standard insulin units (100 IU = 1 mL). This format is optimized for low-volume, high-precision aliquoting of reconstituted peptide solutions in research settings. A separate larger-bore constitution syringe is included for transferring bacteriostatic water from the diluent vial into lyophilized peptide vials, preserving the precision syringes for measurement draws only.
Can the Research Kit 30 Pack be used for compounds other than peptides?
While the Research Kit 30 Pack is optimized for peptide reconstitution workflows, its components are general-purpose sterile laboratory consumables suitable for any aqueous reconstitution requiring a preserved diluent. The alcohol prep pads and syringes are standard research-grade supplies; the bacteriostatic water is appropriate for any compound documented as compatible with 0.9% benzyl alcohol preservative. Compounds requiring non-aqueous solvents (e.g., DMSO, ethanol, acetic acid for certain hydrophobic peptides) will need supplementary diluents not included in this kit.
How does the Research Kit 30 Pack compare to buying components separately?
Purchasing the Research Kit 30 Pack provides a matched 30:30:30 ratio of syringes, prep pads, and bacteriostatic water draws (1 mL per draw from the 30 mL vial) along with a dedicated constitution syringe, eliminating inventory mismatches that commonly disrupt mid-study workflows. Separate sourcing typically results in over- or under-stocking one component and introduces lot-traceability gaps. The kit format also reduces per-unit packaging waste and ensures all components ship together with consistent storage conditions, simplifying receiving and inventory documentation in laboratory settings.
What is the benzyl alcohol concentration in the bacteriostatic water from the Research Kit 30 Pack?
The bacteriostatic water included in the Research Kit 30 Pack contains 0.9% (w/v) benzyl alcohol as the bacteriostatic preservative, dissolved in sterile water for injection. Benzyl alcohol (CAS 100-51-6, MW 108.14 g/mol) at this concentration is the USP-standard preservative loading and provides bacteriostatic activity against a broad range of gram-positive and gram-negative organisms commonly introduced through repeated septum punctures. The 0.9% concentration is sub-cytotoxic at typical reconstitution dilutions used for peptide research and is compatible with the vast majority of peptide structures. Researchers working with neonatal models or compounds known to be sensitive to alcohols should select sterile water instead, as benzyl alcohol can affect certain assay readouts.
Does the Research Kit 30 Pack include needles separately, or are they attached to the syringes?
The 30 insulin-style syringes in the Research Kit 30 Pack feature integrated (fixed) needles — the needle is permanently attached to the syringe barrel as a single low-dead-space unit. This configuration is preferred for sub-milliliter aliquoting because it minimizes residual hub volume to less than 5 µL per draw, compared with 70–80 µL typical of detachable luer-needle configurations. The kit also includes one separate constitution syringe (3 mL luer-slip) with a removable transfer needle, used for the initial drawing of bacteriostatic water from the 30 mL vial into the lyophilized peptide vial. After reconstitution, all subsequent aliquoting is performed using the integrated-needle insulin syringes.
Can the bacteriostatic water in the Research Kit 30 Pack be used to reconstitute lyophilized peptides multiple times from the same vial?
Yes — the 0.9% benzyl alcohol preservative in the kit's bacteriostatic water is specifically formulated for multi-dose vial use, providing antimicrobial protection across repeated septum punctures. USP guidelines for multi-dose preserved diluents support use for up to 28 days after first puncture when stored at 2–8°C, provided aseptic technique is followed at each access. The 30 mL volume in a single vial is sufficient to reconstitute and aliquot multiple lyophilized peptide vials over a typical research timeframe. Each access should be preceded by septum disinfection with a fresh alcohol prep pad (also included in the kit) and a 15-second contact time before needle penetration to maintain the bacteriostatic margin of safety.
Is the Research Kit 30 Pack sterile, and how is sterility maintained between uses?
Each component of the Research Kit 30 Pack is individually packaged to maintain sterility until point of use. The insulin-style syringes are sealed in individual blister packs with sterilized barrels and needles, the alcohol prep pads are foil-sealed to preserve the 70% isopropyl alcohol saturation, and the bacteriostatic water vial is sealed under sterile conditions with a rubber septum that permits multiple punctures while the 0.9% benzyl alcohol preservative inhibits microbial growth during the 28-day in-use period. Between uses, the bacteriostatic water vial should be stored at 2-8°C with the septum disinfected with a fresh prep pad prior to each puncture to prevent contamination.
Can the Research Kit 30 Pack be used in a BSL-1 or BSL-2 laboratory setting?
The Research Kit 30 Pack contains components commonly used in BSL-1 and BSL-2 laboratory workflows for in vitro research applications, including peptide reconstitution for cell culture studies, receptor binding assays, and analytical chemistry preparations. The syringes, prep pads, and bacteriostatic water meet standard laboratory consumable specifications. However, the kit is supplied for research use only and is not certified for clinical, diagnostic, or in vivo administration. Laboratories should follow their institutional biosafety protocols, including appropriate sharps disposal in approved containers and disposal of preservative-containing diluents per local hazardous waste regulations.
What is the shelf life of the unopened Research Kit 30 Pack components?
Unopened components of the Research Kit 30 Pack typically carry shelf lives of 24-36 months from manufacture when stored at controlled room temperature (15-25°C) away from direct sunlight. The bacteriostatic water vial retains its 0.9% benzyl alcohol preservative concentration within compendial specifications throughout this period, while the sealed insulin syringes and foil-pouched alcohol prep pads remain sterile until package integrity is compromised. Expiration dates are printed on individual component packaging. Once the bacteriostatic water vial is first punctured, USP guidance limits in-use stability to 28 days under refrigeration regardless of the unopened shelf life.
What needle gauge and length are the syringes in the Research Kit 30 Pack?
The Research Kit 30 Pack includes insulin-style syringes with 1 mL barrels graduated to 100 units (U-100), typically fitted with 29G to 31G fixed needles measuring 8 mm (5/16 inch) in length. This configuration provides 2-unit (0.02 mL) measurement resolution, suitable for accurate sub-milligram aliquoting of reconstituted peptides at standard research concentrations of 1–10 mg/mL. Fixed-needle insulin syringes also minimize dead-space loss compared to detachable-needle configurations, conserving peptide material across the 30-vial-entry workflow.
How does the benzyl alcohol in the kit's bacteriostatic water affect peptide stability?
At 0.9% (9 mg/mL), benzyl alcohol functions as a bacteriostatic preservative without significantly affecting the chemical stability of most research peptides over the 28-day post-reconstitution window defined by USP . Benzyl alcohol does not cleave peptide bonds and is compatible with the majority of lyophilized peptides studied in published reconstitution literature. Exceptions include certain peptides where benzyl alcohol-induced aggregation has been reported at concentrations above clinical norms; in such cases, plain sterile water with same-day use is preferred. For the kit's intended use across standard research peptides such as BPC-157, TB-500, semaglutide, and ipamorelin, the 0.9% benzyl alcohol concentration is well-established as compatible.
Is the Research Kit 30 Pack compliant with USP standards for laboratory compounding?
The bacteriostatic water in the Research Kit 30 Pack conforms to the USP monograph for Bacteriostatic Water for Injection, including the 0.9% benzyl alcohol preservative specification, sterile filtration (0.22 µm), and endotoxin limits (<0.25 EU/mL) per USP . The syringes meet ISO 8537 standards for sterile insulin syringes, and the alcohol prep pads align with USP antiseptic recommendations using 70% isopropyl alcohol. The kit components individually meet pharmacopeial standards relevant to multi-dose preserved diluents, including the 28-day beyond-use date defined in USP for opened preserved containers stored at 2–8 °C.
Can the Research Kit 30 Pack be used to prepare reference standards for HPLC or LC-MS analysis?
The kit components are well-suited for preparing peptide working solutions for HPLC and LC-MS analysis, though several considerations apply. Bacteriostatic water with 0.9% benzyl alcohol introduces a UV-absorbing component at approximately 257 nm, which may interfere with low-wavelength UV-Vis detection methods; researchers performing absorbance-based quantification should either dilute extensively or substitute LC-MS-grade water for analytical standards. For mass spectrometry workflows, benzyl alcohol elutes early under reverse-phase conditions and typically does not co-elute with peptide analytes. The included syringes and aseptic technique support contamination-free preparation, but final analytical dilutions should use LC-MS-grade solvents in sealed glass vials rather than the kit's polypropylene syringes for maximum analytical reproducibility.
How should used syringes from the Research Kit 30 Pack be disposed of?
Used insulin syringes from the Research Kit 30 Pack should be disposed of in an FDA-cleared, puncture-resistant sharps container immediately after use, following OSHA Bloodborne Pathogens Standard (29 CFR 1910.1030) and CDC guidelines. Never recap, bend, or manually manipulate used needles, as recapping is the leading cause of needlestick injury. Sharps containers should be filled to no more than 75% capacity and disposed of through a licensed regulated medical waste (RMW) hauler or, in jurisdictions that permit it, an approved mail-back program. Place the sharps container within arm's reach of the work area to minimize transit with exposed needles.
Can the syringes in the Research Kit 30 Pack be reused on the same vial?
No — each insulin syringe in the Research Kit 30 Pack is designed for single-use only, even when drawing from the same multi-dose vial. Reusing syringes introduces several risks: needle dulling after the first septum penetration increases coring and rubber particulate contamination on subsequent draws, plunger lubricant degradation alters dosing accuracy, and any external contamination of the needle becomes a contamination vector for the vial contents. The 30-syringe count in the kit is matched to the 30 mL bacteriostatic water volume specifically to support a one-syringe-per-draw workflow over the 28-day BAC water in-use stability window.
Are the alcohol prep pads in the Research Kit 30 Pack sterile?
Yes — the alcohol prep pads in the Research Kit 30 Pack are individually packaged, sterile-by-saturation pads containing 70% isopropyl alcohol (IPA), the concentration established by USP and CDC guidelines as optimal for surface antisepsis. The 70% concentration is more effective than higher concentrations (90%+ IPA) because water content slows evaporation, allowing sufficient contact time to denature microbial proteins. Each pad is sealed in a foil-laminate pouch that maintains sterility and prevents IPA evaporation until the moment of use. Pads should be used immediately after opening, as exposed IPA evaporates within 30-60 seconds and loses antiseptic efficacy.
What happens if the bacteriostatic water in the Research Kit 30 Pack freezes during shipping?
If the bacteriostatic water (BAC water) component freezes during winter shipping, allow the vial to thaw completely at room temperature (20-25°C) before use and inspect the solution visually. Freezing of 0.9% benzyl alcohol bacteriostatic water does not chemically degrade the preservative or the water itself, and stability data indicate the solution remains usable after a single freeze-thaw cycle provided the vial seal is intact and the solution is clear, colorless, and free of particulates after thawing. However, repeated freeze-thaw cycles may stress the rubber septum and compromise seal integrity. If the vial shows cloudiness, precipitate, or a compromised seal after thawing, it should be discarded. Lyophilized peptides reconstituted with previously frozen BAC water show no documented loss of potency in published stability studies.
What volume of bacteriostatic water is included in the Research Kit 30 Pack, and how many reconstitutions does it support?
The Research Kit 30 Pack includes 30 mL of bacteriostatic water containing 0.9% benzyl alcohol as a preservative. This volume supports approximately 10-30 individual peptide vial reconstitutions depending on the target concentration. For example, reconstituting a 5 mg lyophilized peptide vial at 5 mg/mL requires 1 mL of diluent, allowing 30 reconstitutions from the kit. At 2 mg/mL working concentration (2.5 mL per 5 mg vial), the same volume supports 12 reconstitutions. The 30 mL format with benzyl alcohol preservative supports the 28-day USP beyond-use date for multi-dose use after initial septum penetration.
Can the constitution syringe in the Research Kit 30 Pack be reused for multiple peptide preparations?
The larger constitution syringe is designed for the initial transfer of bacteriostatic water from the diluent vial to the lyophilized peptide vial. While it can technically be reused on the same diluent vial across multiple reconstitution sessions, best practice dictates single-use to maintain sterility and prevent cross-contamination between peptide preparations. If reusing within a short session (e.g., reconstituting multiple vials of the same peptide consecutively), the syringe should remain capped between uses, the diluent vial septum should be re-disinfected with an alcohol prep pad before each penetration, and the syringe should be discarded into a sharps container at session end.
Are the syringes in the Research Kit 30 Pack latex-free and suitable for sensitive compound research?
The insulin syringes included in the Research Kit 30 Pack are manufactured to standard laboratory specifications using polypropylene barrels, polyethylene plungers with synthetic rubber tips, and stainless steel needles. Modern medical-grade insulin syringes are typically latex-free to comply with healthcare and laboratory safety standards, eliminating concerns about latex protein contamination of sensitive peptide preparations or interference with downstream assays. Plunger tips use synthetic elastomers (commonly silicone-coated isoprene or thermoplastic elastomer) that exhibit low extractables and leachables profiles, supporting compatibility with most aqueous peptide solutions for short-duration handling.
How does the Research Kit 30 Pack support reproducibility in peptide research workflows?
The Research Kit 30 Pack supports experimental reproducibility by providing standardized, lot-traceable components for every reconstitution event in a study. Using consistent syringe dead space (~1-3 µL), uniform 70% isopropyl alcohol prep pad disinfection, and a single 30 mL bacteriostatic water source with documented 0.9% benzyl alcohol concentration eliminates batch-to-batch variability that can arise when sourcing components separately. This consistency is particularly relevant for dose-response studies, pharmacokinetic analyses, and any research where small variations in concentration (±5%) could confound results. The bundled format also simplifies materials documentation in laboratory notebooks and method sections of research publications.
Can the Research Kit 30 Pack be used for in vitro cell culture peptide preparations?
The bacteriostatic water in the Research Kit 30 Pack contains 0.9% benzyl alcohol as a preservative, which can be cytotoxic to certain sensitive cell lines — particularly primary neonatal cultures, neuronal models, and some stem cell lines — at concentrations above approximately 0.003%. For in vitro applications where benzyl alcohol interference is a concern, sterile water for injection or cell-culture-grade diluents are recommended instead. For routine adult cell line work where the final benzyl alcohol concentration after dilution falls below 0.001%, the kit's bacteriostatic water remains suitable. The syringes, prep pads, and constitution syringe components are appropriate for any sterile liquid handling workflow.
What is the recommended needle insertion angle for the syringes in the Research Kit 30 Pack?
Peer-reviewed septum integrity research supports a 45° insertion angle with the needle bevel oriented upward for minimizing rubber coring during vial septum penetration. Studies have shown that 45° bevel-up insertion reduces coring incidence by approximately 62% compared to perpendicular (90°) insertion across needle gauges. The fine 28-30 gauge needles of insulin-style syringes in the Research Kit 30 Pack inherently produce very low coring rates (under 0.8% across 100 penetrations), but using the recommended angled technique further preserves septum integrity for vials accessed repeatedly across multi-day or multi-week protocols.
Does the Research Kit 30 Pack include documentation for laboratory traceability?
Each Research Kit 30 Pack ships with lot-level traceability information for the bacteriostatic water and syringe components, supporting documentation requirements for laboratories operating under quality management frameworks such as ISO 17025, GLP-aligned protocols, or institutional research compliance standards. The bacteriostatic water is sourced from manufacturers compliant with USP monograph standards for Bacteriostatic Water for Injection, including 0.9% w/v benzyl alcohol preservative content and sterility testing. Researchers requiring Certificates of Analysis for specific lots should request these through AminoCore Research customer support prior to placing the order so the appropriate documentation can be matched to the shipped lots.
How does the Research Kit 30 Pack support reproducibility across multi-week experimental campaigns?
Reproducibility in peptide research depends on consistent diluent composition, syringe dead-space volumes, and aseptic technique across every reconstitution and dose withdrawal event in an experiment. The Research Kit 30 Pack addresses this by supplying all 30 reconstitution and dosing consumables from a single matched lot set — eliminating the inter-lot variability that occurs when sourcing syringes, prep pads, and diluent separately from different vendors across the course of a multi-week study. This standardization reduces a known source of analytical variability in HPLC quantification, dose-response curve fitting, and time-course pharmacokinetic modeling, making the kit particularly well-suited for longitudinal research designs where consumable consistency is a documented confounding variable.
What endotoxin levels are present in the bacteriostatic water from the Research Kit 30 Pack?
The bacteriostatic water in the Research Kit 30 Pack is manufactured to USP standards for Bacteriostatic Water for Injection, which specifies an endotoxin limit of ≤0.5 EU/mL by Limulus Amebocyte Lysate (LAL) assay. In practice, USP-compliant bacteriostatic water typically measures
How accurate are the insulin syringes in the Research Kit 30 Pack for low-volume peptide dosing?
The 1 mL U-100 insulin syringes in the Research Kit 30 Pack are manufactured to ISO 8537:2016 standards, which specify ±5% volumetric accuracy at nominal fill volume and ±10% accuracy at volumes ≤10% of nominal. Each unit graduation represents 0.01 mL (10 µL), providing practical resolution for typical research-scale aliquots. To maintain optimal accuracy, researchers should plan reconstitution volumes that produce draw volumes of at least 10 units (0.10 mL). The fixed-needle design also minimizes dead space to approximately 1-3 µL, an order of magnitude lower than detachable Luer-lock syringes, which improves dose recovery for high-value peptides.
What happens to the Research Kit 30 Pack components during shipping temperature excursions?
The Research Kit 30 Pack tolerates transient shipping excursions across approximately 2-40°C without performance loss. Benzyl alcohol-preserved bacteriostatic water remains chemically stable in this window, though brief freezing should prompt inspection of the vial seal, septum, and contents for cracking or crystalline residue after thawing. Sustained exposure above 40°C can accelerate benzyl alcohol oxidation—any yellow discoloration indicates degradation and the vial should be discarded. Insulin syringe pouches and foil-wrapped alcohol prep pads maintain sterility provided packaging remains intact; flat prep pad packets indicate alcohol evaporation through a compromised seal and reduced antiseptic efficacy.
Is the Research Kit 30 Pack suitable for preparing peptides for cell culture experiments?
Yes, the Research Kit 30 Pack is suitable for in vitro cell culture peptide preparation, with several important considerations. The USP-grade bacteriostatic water provides low-endotoxin diluent (typically
Can the Research Kit 30 Pack be used for preparing peptide solutions for animal model studies?
The Research Kit 30 Pack is designed for laboratory research applications and provides the sterile consumables required for aseptic reconstitution of lyophilized peptides used in in vitro and in vivo preclinical workflows. The 0.9% benzyl alcohol bacteriostatic water is widely cited in animal model research for reconstituting multi-dose peptide stocks, with the caveat that benzyl alcohol has documented toxicity in neonatal rodent models and should be avoided in studies involving very young animals. For adult rodent or larger animal model work, the kit's components meet the sterility, volumetric accuracy, and contamination-control standards required for reproducible dosing. Always consult institutional IACUC protocols and peptide-specific stability data before deployment.
How does the Research Kit 30 Pack mitigate cross-contamination between different peptide stock vials?
The kit contains 30 individually wrapped single-use insulin syringes, allowing researchers to assign one syringe per vial per session — a discipline that eliminates the highest-impact contamination vector identified in laboratory reconstitution workflows. Combined with the 30 alcohol prep pads (one per septum penetration event), the kit supports a one-vial-one-syringe-one-pad protocol that prevents needle-mediated transfer of residual material or microbial inoculum between stocks. This is particularly important when working with multiple peptides in the same session, where carryover at the trace level can introduce analytical variability in downstream HPLC or LC-MS characterization.
Should reconstituted peptide vials prepared with the Research Kit 30 Pack be protected from light?
Yes, for peptides containing photosensitive residues including tryptophan, tyrosine, methionine, and cysteine. Once reconstituted with the kit's bacteriostatic water, peptide solutions become susceptible to photochemical degradation pathways that are largely absent in the lyophilized state. Even ambient laboratory fluorescent lighting can drive measurable methionine oxidation over multi-day storage windows. Reconstituted vials should be returned to refrigerated storage (2–8 °C) in an opaque container or amber-wrapped vial immediately after each dose withdrawal. The bacteriostatic preservative system prevents microbial growth but does not protect against oxidative or photochemical degradation.
What documentation should accompany each reconstitution event when using the Research Kit 30 Pack?
Reproducible peptide research requires capturing the lot numbers of all kit components used (bacteriostatic water, syringe lot, prep pad lot), the peptide vial identity (supplier, lot, COA reference), the reconstitution calculation (peptide mass, diluent volume, final concentration), and the reconstitution date with operator initials. Each reconstituted vial should be labeled with peptide name, concentration in mg/mL, reconstitution date, expiration date based on stability data, and diluent type. Per-dose withdrawal logs should record date, time, volume, calculated dose mass, and any anomalies observed. This documentation chain enables forward and backward traceability for audit reconstruction and supports GLP-compatible workflows.
What is the difference between the insulin syringes and the constitution syringe in the Research Kit 30 Pack?
The 30 individually wrapped insulin syringes in the Research Kit 30 Pack are 1 mL (or smaller) U-100 graduated syringes with fixed 29G–31G needles, designed for precise low-volume aliquoting of reconstituted peptide solutions. The single constitution syringe (also called a mixing syringe) is a larger-volume syringe — typically 3 mL or 5 mL — used to transfer bacteriostatic water from the diluent vial into the lyophilized peptide vial during the initial reconstitution step. Separating these functions reduces cross-contamination risk, since the constitution syringe contacts only the diluent vial and one peptide vial septum, while each insulin syringe is used once for a single dose aliquot and discarded.
Can the Research Kit 30 Pack be used to prepare peptide stock solutions for long-term frozen storage?
The Research Kit 30 Pack can be used to reconstitute lyophilized peptides into aqueous stock solutions, which may then be aliquoted into separate cryovials for frozen storage at −20°C or −80°C. However, researchers should note that the bacteriostatic water diluent contains 0.9% benzyl alcohol, which can co-precipitate or crystallize on freezing and may not be ideal for downstream applications sensitive to preservative carry-over (e.g., cell culture, in vivo dosing in benzyl alcohol–sensitive species). For long-term frozen archival stocks, sterile water for injection or a peptide-appropriate buffer is often preferred. The kit's bacteriostatic water is optimized for short- to medium-term refrigerated multi-dose use rather than deep-frozen archives.
Are the syringes in the Research Kit 30 Pack appropriate for viscous peptide solutions or oil-based vehicles?
The insulin-style 29G–31G syringes in the Research Kit 30 Pack are optimized for low-viscosity aqueous solutions, which matches the profile of most peptide reconstitutions in bacteriostatic water. High-viscosity preparations — such as peptides formulated in propylene glycol, DMSO–water mixtures, or oil-based research vehicles — generate significant flow resistance through fine-gauge needles and may require excessive plunger force, leading to volumetric inaccuracy or needle hub disconnection. For viscous vehicles, larger-bore needles (21G–23G) and Luer-lock syringes are more appropriate. The kit components are best suited to standard aqueous peptide reconstitutions at concentrations typical of laboratory stock solutions (0.5–10 mg/mL).
How does the Research Kit 30 Pack support Good Laboratory Practice (GLP) documentation requirements?
While the Research Kit 30 Pack itself is not GLP-certified, its components carry individual lot numbers (bacteriostatic water vial, syringe blister packs, prep pad sachets) that support GLP-style chain-of-custody documentation. Researchers operating under GLP, ISO/IEC 17025, or institutional reproducibility protocols should record kit component lot numbers, opening dates, and reconstitution event details in their experimental notebooks or LIMS. This enables retrospective traceability if a contamination event, potency drift, or unexpected experimental result is investigated. The kit's standardized component set also reduces protocol variability across operators and experimental campaigns, which is a foundational element of GLP-compliant workflows.
What personal protective equipment is recommended when using the Research Kit 30 Pack?
Baseline PPE for laboratory peptide reconstitution with the Research Kit 30 Pack includes powder-free nitrile gloves (4-mil minimum), a long-sleeved laboratory coat or disposable gown, and side-shielded safety eyewear. For lyophilised peptide vial opening or solid compound weighing, surgical or N95 respiratory protection is advised, ideally within a certified biosafety cabinet or chemical fume hood. Gloves should be changed between different compound vials to prevent cross-contamination, and hands washed with soap and water immediately after glove removal. The kit components themselves are latex-free, but researchers with sensitivities to benzyl alcohol (0.9% in bacteriostatic water) or isopropyl alcohol (in prep pads) should review safety data before use.
Can silicone oil from the syringes in the Research Kit 30 Pack affect peptide stability?
Insulin-style syringes are lubricated with medical-grade silicone oil (polydimethylsiloxane) on the inner barrel wall. Studies using flow imaging microscopy have detected 1,000-5,000 silicone oil droplets per mL in solutions drawn from siliconised syringes, predominantly in the 2-10 μm range. For most peptides, single-use draw-up has negligible impact on stability. However, hydrophobic peptides (e.g., lipidated GLP-1 agonists, fatty-acid-conjugated compounds) can adsorb to oil droplets, with agitation increasing particle counts 3-7 fold. To minimise impact, the syringes in the Research Kit 30 Pack should be used for immediate dosing rather than as storage containers; reconstituted peptide should remain in the original glass vial, with fresh aliquots drawn only at the point of use.
How should reconstitution volume be calculated when using the Research Kit 30 Pack with different peptide vial sizes?
Reconstitution volume selection balances dosing precision against syringe capacity. For a 5 mg lyophilised vial reconstituted with 2 mL of bacteriostatic water from the Research Kit, final concentration is 2.5 mg/mL — a 250 μg dose requires 0.10 mL (10 units on a 100-unit insulin syringe). For 10 mg vials, 2 mL reconstitution yields 5 mg/mL (500 μg per 0.10 mL); 3 mL yields 3.33 mg/mL with larger dose volumes and reduced relative error. The 30 mL of bacteriostatic water in the kit supports approximately 15 vial reconstitutions at 2 mL per vial. Gravimetric verification (weighing pre- and post-diluent addition; water = 1.00 g/mL at 20°C) provides an audit trail for GLP-compliant workflows.
Can the Research Kit 30 Pack be used for compounds requiring serial dilutions or concentration-response studies?
Yes. The 30 mL of bacteriostatic water and 30 insulin syringes in the Research Kit 30 Pack support serial dilution workflows commonly used in concentration-response studies. The recommended approach is to reconstitute the master peptide vial at a moderate concentration (e.g., 2.5-5 mg/mL), then prepare working dilutions in separate sterile vials using fresh syringes and additional bacteriostatic water. This preserves the integrity of the master stock by limiting septum penetrations and minimises cross-contamination between dilution levels. For half-log or log dilution series, gravimetric verification of dilution volumes is recommended to maintain precision. Note that the bacteriostatic water diluent contains 0.9% benzyl alcohol, which is suitable for most aqueous peptide dilutions but may not be appropriate for cell culture work where preservative-free sterile water is preferred.
Can peptides adsorb onto the syringes in the Research Kit 30 Pack and reduce dosing accuracy?
Peptide adsorption onto polypropylene syringe barrels is concentration-dependent and most pronounced below 10 µg/mL. At typical reconstitution concentrations of 1–10 mg/mL used with the Research Kit 30 Pack, adsorption losses are functionally negligible (
What is the USP standard for the bacteriostatic water in the Research Kit 30 Pack?
The bacteriostatic water for injection (BWFI) supplied with the Research Kit 30 Pack conforms to the USP monograph specifying sterile water with 0.9% (9 mg/mL) benzyl alcohol as antimicrobial preservative, endotoxin limit ≤0.25 EU/mL, and pH 4.5–7.0. This compendial standard supports the widely cited 28-day beyond-use date for reconstituted peptide vials stored at 2–8°C when aseptic technique is maintained. Benzyl alcohol at this concentration meets USP Antimicrobial Effectiveness Testing Category 1 requirements against S. aureus, E. coli, P. aeruginosa, C. albicans, and A. brasiliensis.
What should I do if the reconstituted peptide solution appears cloudy after using the Research Kit 30 Pack?
Cloudy or turbid solutions after reconstitution typically indicate peptide aggregation, often caused by vigorous shaking, incompatible pH, or excessive concentration. The recommended corrective action is to discard the affected vial and reconstitute a fresh aliquot using gentle swirling rather than vortexing or shaking, and to lower the target concentration to 1–2 mg/mL. Some hydrophobic peptides require acidified diluents (0.1% acetic acid) or co-solvent pre-dissolution before transfer to BWFI from the Research Kit 30 Pack. Always consult peptide-specific solubility data before reconstitution.
How does the Research Kit 30 Pack support GLP-aligned reconstitution documentation?
The Research Kit 30 Pack supports Good Laboratory Practice documentation by providing traceable, lot-numbered components (BWFI, syringes, prep pads) that can be recorded in laboratory notebooks or electronic records at each reconstitution event. GLP-aligned documentation typically captures the kit lot number, peptide vial identifier, reconstitution date and time, calculated concentration, operator initials, and beyond-use date assigned per USP guidance (28 days at 2–8°C for BWFI-reconstituted vials). Maintaining this chain-of-custody record for every kit-prepared vial satisfies traceability and reproducibility requirements for multi-week experimental campaigns.
Can the Research Kit 30 Pack be used inside a biosafety cabinet for sterile reconstitution work?
Yes. The components of the Research Kit 30 Pack — sterile insulin syringes, individually wrapped 70% isopropyl alcohol prep pads, bacteriostatic water in a sealed multi-dose vial, and the constitution syringe — are all compatible with Class II biosafety cabinet (BSC) workflows. Using the kit inside a BSC provides ISO Class 5 HEPA-filtered laminar airflow at the work surface, which substantially reduces airborne particulate and microbial deposition compared to open-bench reconstitution. Investigators performing reconstitution for cell culture, in vitro assays, or animal studies are encouraged to use a BSC where available, and to allow the cabinet to run for at least 5 minutes before introducing materials. Surface-wipe the BSC interior with 70% isopropyl alcohol before staging the kit components.
What is the dead-space volume of the insulin syringes in the Research Kit 30 Pack, and how does it affect dosing accuracy?
The insulin syringes in the Research Kit 30 Pack feature integrated (fixed) needles, which produce a dead-space volume of approximately 1-3 microlitres — substantially lower than detachable hub-and-needle configurations, which can retain 35-85 microlitres. This low dead space minimises peptide loss during dose preparation, preserving more than 99.9% of the intended dose volume in typical 0.1-0.5 mL research applications. For a 5 mg/mL peptide solution drawn at 0.2 mL, the maximum theoretical dose retention loss is approximately 15 micrograms, compared to 175-425 micrograms with high-dead-space syringes. This engineering feature supports both dose precision and economical use of expensive research compounds across multi-vial workflows.
Are the components in the Research Kit 30 Pack tested for endotoxin or pyrogen content?
The bacteriostatic water component in the Research Kit 30 Pack is manufactured to USP Water for Injection standards with subsequent addition of 0.9% benzyl alcohol, and is tested by the manufacturer for bacterial endotoxin content using the Limulus Amebocyte Lysate (LAL) assay per USP . Typical endotoxin levels are below 0.25 EU/mL, well within the compendial limit of 0.5 EU/mL for sterile water for injection. The syringes and prep pads are supplied as sterile single-use devices but are not individually endotoxin-tested at the lot level for laboratory research applications. Investigators performing endotoxin-sensitive assays (e.g., macrophage activation studies, TLR4 signalling experiments) should consider supplementary endotoxin testing of their final reconstituted solutions.
Does the Research Kit 30 Pack include instructions or a reconstitution worksheet?
The Research Kit 30 Pack is supplied as a component kit and does not include compound-specific reconstitution instructions, since the appropriate diluent volume and resulting concentration depend on the peptide identity, mass per vial, and intended experimental dose. AminoCore Research provides detailed reconstitution guidance on each individual peptide product page, including recommended diluent volumes, target concentrations, and storage parameters for the reconstituted solution. Investigators are encouraged to print and complete a reconstitution worksheet documenting the date, lot numbers of both the peptide and bacteriostatic water, diluent volume added, final concentration, operator initials, and storage location — supporting Good Laboratory Practice (GLP) traceability across multi-week experimental campaigns.
For laboratory and research use only. Not intended for human or animal consumption. All product information is derived from published preclinical research and does not constitute medical advice or claims.